Omics Signatures of Tissue Injury and Hemorrhagic Shock in Swine
- PMID: 37334680
- PMCID: PMC10728352
- DOI: 10.1097/SLA.0000000000005944
Omics Signatures of Tissue Injury and Hemorrhagic Shock in Swine
Abstract
Objective: Advanced mass spectrometry methods were leveraged to analyze both proteomics and metabolomics signatures in plasma upon controlled tissue injury (TI) and hemorrhagic shock (HS)-isolated or combined-in a swine model, followed by correlation to viscoelastic measurements of coagulopathy via thrombelastography.
Background: TI and HS cause distinct molecular changes in plasma in both animal models and trauma patients. However, the contribution to coagulopathy of trauma, the leading cause of preventable mortality in this patient population remains unclear. The recent development of a swine model for isolated or combined TI+HS facilitated the current study.
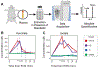
Methods: Male swine (n=17) were randomized to either isolated or combined TI and HS. Coagulation status was analyzed by thrombelastography during the monitored time course. The plasma fractions of the blood draws (at baseline; end of shock; and at 30 minutes, 1, 2, and 4 hours after shock) were analyzed by mass spectrometry-based proteomics and metabolomics workflows.
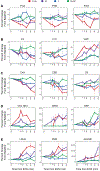
Results: HS-isolated or combined with TI-caused the most severe omic alterations during the monitored time course. While isolated TI delayed the activation of coagulation cascades. Correlation to thrombelastography parameters of clot strength (maximum amplitude) and breakdown (LY30) revealed signatures of coagulopathy which were supported by analysis of gene ontology-enriched biological pathways.
Conclusion: The current study provides a comprehensive characterization of proteomic and metabolomic alterations to combined or isolated TI and HS in a swine model and identifies early and late omics correlates to viscoelastic measurements in this system.
Copyright © 2023 Wolters Kluwer Health, Inc. All rights reserved.
Conflict of interest statement
A.D. and K.C.H. are founders of Omix Technologies Inc. A.D. is also a founder of Altis Biosciences LLC., A.D. is a consultant for Macopharma Inc. A.D. and C.C.S. are both consultants for Hemanext Inc. E.E.M. has received research support from Haemonetics, Werfen, Hemosonics, Stago, Diapharma, and Prytime. The remaining authors report no conflicts of interest.
Figures





References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

