Effects of selective cyclooxygenase-2 inhibitor robenacoxib on primary cells derived from feline injection-site sarcoma
- PMID: 37334757
- PMCID: PMC10399534
- DOI: 10.1111/jcmm.17717
Effects of selective cyclooxygenase-2 inhibitor robenacoxib on primary cells derived from feline injection-site sarcoma
Abstract
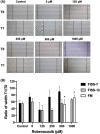
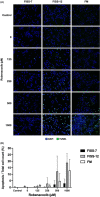
Feline injection-site sarcomas (FISSs) are highly invasive malignant mesenchymal neoplasms that arise from injection sites in cats. Although the tumorigenesis of FISSs is still uncertain, there is a consensus that FISS is associated with chronic inflammation caused by irritation of injection-related trauma and foreign chemical substances. Chronic inflammation can provide a proper microenvironment for tumour development, which has been known as one of the risk factors of tumorigenesis in many tumours. To investigate the tumorigenesis of FISS and screen for its potential therapeutic targets, cyclooxygenase-2 (COX-2), an inflammation-enhancing enzyme, was selected as a target for this study. In vitro experiments using FISS- and normal tissue-derived primary cells and robenacoxib, a highly selective COX-2 inhibitor, were performed. The results demonstrated that expression of COX-2 could be detected in formalin-fixed and paraffin-embedded FISS tissues and FISS-derived primary cells. Cell viability, migration and colony formation of FISS-derived primary cells were inhibited, and cell apoptosis was enhanced by robenacoxib in a dose-dependent manner. However, susceptibility to robenacoxib varied in different lines of FISS primary cells and was not completely correlated with COX-2 expression. Our results suggest that COX-2 inhibitors could be potential adjuvant therapeutics against FISSs.
Keywords: cyclooxygenase-2; feline injection-site sarcoma; primary cells; robenacoxib.
© 2023 The Authors. Journal of Cellular and Molecular Medicine published by Foundation for Cellular and Molecular Medicine and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




Similar articles
-
Investigating the role of signal transducer and activator of transcription 3 in feline injection site sarcoma.BMC Vet Res. 2022 Jul 14;18(1):276. doi: 10.1186/s12917-022-03352-y. BMC Vet Res. 2022. PMID: 35836213 Free PMC article.
-
Comparison of MMP-2, MMP-9, COX-2, and PGP Expression in Feline Injection-Site and Feline Noninjection-Site Sarcomas-Pilot Study.Animals (Basel). 2024 Jul 19;14(14):2110. doi: 10.3390/ani14142110. Animals (Basel). 2024. PMID: 39061572 Free PMC article.
-
Epidemiological and morphological analysis of feline injection site sarcomas.Pol J Vet Sci. 2015;18(2):313-22. doi: 10.1515/pjvs-2015-0041. Pol J Vet Sci. 2015. PMID: 26172181
-
Pharmacology, safety, efficacy and clinical uses of the COX-2 inhibitor robenacoxib.J Vet Pharmacol Ther. 2022 Jul;45(4):325-351. doi: 10.1111/jvp.13052. Epub 2022 Apr 22. J Vet Pharmacol Ther. 2022. PMID: 35460083 Free PMC article. Review.
-
Feline injection-site sarcoma: past, present and future perspectives.Vet J. 2011 May;188(2):136-41. doi: 10.1016/j.tvjl.2010.04.025. Epub 2010 May 26. Vet J. 2011. PMID: 20510635 Review.
References
-
- Hendrick M, Goldschmidt M. Do injection site reactions induce fibrosarcomas in cats? J Am Vet Med Assoc. 1991;199(8):968. - PubMed
-
- Yeh JL‐S, Chou T‐H, Hou S‐Y. Evolving terminology for injection‐site sarcomas in cats. J Am Vet Med Assoc. 2016;249(7):738.
-
- Kass PH, Barnes W Jr, Spangler W, Chomel BB, Culbertson M. Epidemiologic evidence for a causal relation between vaccination and fibrosarcoma tumorigenesis in cats. J Am Vet Med Assoc. 1993;203(3):396‐405. - PubMed
-
- Hendrick M, Brooks J. Postvaccinal sarcomas in the cat: histology and immunohistochemistry. Vet Pathol. 1994;31(1):126‐129. - PubMed
-
- Esplin D, Bigelow M, McGill L, Wilson S. Fibrosarcoma at the site of a lufenuron injection in a cat. Vet Cancer Soc Newsletter. 1999;23(2):8‐9.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

