Peripheral BDNF Regulates Somatosensory-Sympathetic Coupling in Brachial Plexus Avulsion-Induced Neuropathic Pain
- PMID: 37335428
- PMCID: PMC10661543
- DOI: 10.1007/s12264-023-01075-0
Peripheral BDNF Regulates Somatosensory-Sympathetic Coupling in Brachial Plexus Avulsion-Induced Neuropathic Pain
Abstract
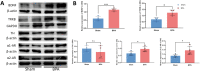
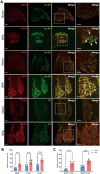
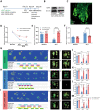
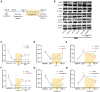
Brachial plexus avulsion (BPA) is a combined injury involving the central and peripheral nervous systems. Patients with BPA often experience severe neuropathic pain (NP) in the affected limb. NP is insensitive to the existing treatments, which makes it a challenge to researchers and clinicians. Accumulated evidence shows that a BPA-induced pain state is often accompanied by sympathetic nervous dysfunction, which suggests that the excitation state of the sympathetic nervous system is correlated with the existence of NP. However, the mechanism of how somatosensory neural crosstalk with the sympathetic nerve at the peripheral level remains unclear. In this study, through using a novel BPA C7 root avulsion mouse model, we found that the expression of BDNF and its receptor TrκB in the DRGs of the BPA mice increased, and the markers of sympathetic nervous system activity including α1 and α2 adrenergic receptors (α1-AR and α2-AR) also increased after BPA. The phenomenon of superexcitation of the sympathetic nervous system, including hypothermia and edema of the affected extremity, was also observed in BPA mice by using CatWalk gait analysis, an infrared thermometer, and an edema evaluation. Genetic knockdown of BDNF in DRGs not only reversed the mechanical allodynia but also alleviated the hypothermia and edema of the affected extremity in BPA mice. Further, intraperitoneal injection of adrenergic receptor inhibitors decreased neuronal excitability in patch clamp recording and reversed the mechanical allodynia of BPA mice. In another branch experiment, we also found the elevated expression of BDNF, TrκB, TH, α1-AR, and α2-AR in DRG tissues from BPA patients compared with normal human DRGs through western blot and immunohistochemistry. Our results revealed that peripheral BDNF is a key molecule in the regulation of somatosensory-sympathetic coupling in BPA-induced NP. This study also opens a novel analgesic target (BDNF) in the treatment of this pain with fewer complications, which has great potential for clinical transformation.
Keywords: Brachial plexus avulsion; Brain-derived neurotrophic factor; Mechanical allodynia; Neuropathic pain; Peripheral sensitization; Sympathetic nervous system.
© 2023. Center for Excellence in Brain Science and Intelligence Technology, Chinese Academy of Sciences.
Conflict of interest statement
All authors declare that there are no conflicts of interest.
Figures




References
-
- Noland SS, Bishop AT, Spinner RJ, Shin AY. Adult traumatic brachial plexus injuries. J Am Acad Orthop Surg. 2019;27:705–716. - PubMed
-
- Rodrigues-Filho R, Santos AR, Bertelli JA, Calixto JB. Avulsion injury of the rat brachial plexus triggers hyperalgesia and allodynia in the hindpaws: A new model for the study of neuropathic pain. Brain Res. 2003;982:186–194. - PubMed
-
- Le W, Liu Y, Zhou Y, Lao J, Zhao X. A new rat model of neuropathic pain: Complete brachial plexus avulsion. Neurosci Lett. 2015;589:52–56. - PubMed
-
- Pace MC, Passavanti MB, De Nardis L, Bosco F, Sansone P, Pota V, et al. Nociceptor plasticity: A closer look. J Cell Physiol. 2018;233:2824–2838. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

