Health Equity in Patients Receiving Durvalumab for Unresectable Stage III Non-Small Cell Lung Cancer in the US Veterans Health Administration
- PMID: 37335901
- PMCID: PMC10485300
- DOI: 10.1093/oncolo/oyad172
Health Equity in Patients Receiving Durvalumab for Unresectable Stage III Non-Small Cell Lung Cancer in the US Veterans Health Administration
Abstract
Background: Real-world evidence is limited regarding the relationship between race and use of durvalumab, an immunotherapy approved for use in adults with unresectable stage III non-small cell lung cancer (NSCLC) post-chemoradiotherapy (CRT). This study aimed to evaluate if durvalumab treatment patterns differed by race in patients with unresectable stage III NSCLC in a Veterans Health Administration (VHA) population.
Materials and methods: This was a retrospective analysis of White and Black adults with unresectable stage III NSCLC treated with durvalumab presenting to any VHA facility in the US from January 1, 2017, to June 30, 2020. Data captured included baseline characteristics and durvalumab treatment patterns, including treatment initiation delay (TID), interruption (TI), and discontinuation (TD); defined as CRT completion to durvalumab initiation greater than 42 days, greater than 28 days between durvalumab infusions, and more than 28 days from the last durvalumab dose with no new durvalumab restarts, respectively. The number of doses, duration of therapy, and adverse events were also collected.
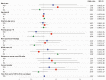
Results: A total of 924 patients were included in this study (White = 726; Black = 198). Race was not a significant factor in a multivariate logistic regression model for TID (OR, 1.39; 95% CI, 0.81-2.37), TI (OR, 1.58; 95% CI, 0.90-2.76), or TD (OR, 0.84; 95% CI, 0.50-1.38). There were also no significant differences in median (interquartile range [IQR]) number of doses (White: 15 [7-24], Black: 18 [7-25]; P = .25) or median (IQR) duration of therapy (White: 8.7 months [2.9-11.8], Black: 9.8 months [3.6-12.0]; P = .08), although Black patients were less likely to experience an immune-related adverse event (28% vs. 36%, P = .03) and less likely to experience pneumonitis (7% vs. 14%, P < .01).
Conclusion: Race was not found to be linked with TID, TI, or TD in this real-world study of patients with unresectable stage III NSCLC treated with durvalumab at the VHA.
Keywords: durvalumab; health disparity; health equity; immunotherapy; lung cancer.
© The Author(s) 2023. Published by Oxford University Press.
Conflict of interest statement
The institutions of Christopher R. Frei, Zohra Nooruddin, Kelly R. Reveles, and Xavier Jones have received grant money, for them to perform research, from AstraZeneca, in the last three years. Lance Brannman, Ion Cotarla, and Tiernan Mulrooney are employees of AstraZeneca Pharmaceuticals. Andrew Frankart was supported by the ASTRO-AstraZeneca Radiation Oncology Research Training Fellowship. The other authors indicated no financial relationships.
Figures
Similar articles
-
Durvalumab Treatment Patterns for Patients with Unresectable Stage III Non-Small Cell Lung Cancer in the Veterans Health Administration (VHA): A Nationwide, Real-World Study.Curr Oncol. 2023 Sep 13;30(9):8411-8423. doi: 10.3390/curroncol30090611. Curr Oncol. 2023. PMID: 37754526 Free PMC article.
-
Durvalumab After Chemoradiotherapy in Patients With Unresectable Stage III Non-Small Cell Lung Cancer.JAMA Netw Open. 2024 Apr 1;7(4):e247542. doi: 10.1001/jamanetworkopen.2024.7542. JAMA Netw Open. 2024. PMID: 38648057 Free PMC article.
-
Treatment Characteristics and Real-World Progression-Free Survival in Patients With Unresectable Stage III NSCLC Who Received Durvalumab After Chemoradiotherapy: Findings From the PACIFIC-R Study.J Thorac Oncol. 2023 Feb;18(2):181-193. doi: 10.1016/j.jtho.2022.10.003. Epub 2022 Oct 25. J Thorac Oncol. 2023. PMID: 36307040
-
Real-World Safety and Efficacy of Consolidation Durvalumab After Chemoradiation Therapy for Stage III Non-small Cell Lung Cancer: A Systematic Review and Meta-analysis.Int J Radiat Oncol Biol Phys. 2022 Apr 1;112(5):1154-1164. doi: 10.1016/j.ijrobp.2021.12.150. Epub 2021 Dec 26. Int J Radiat Oncol Biol Phys. 2022. PMID: 34963558
-
Immune checkpoint-inhibitors and chemoradiation in stage III unresectable non-small cell lung cancer.Lung Cancer. 2019 Aug;134:259-267. doi: 10.1016/j.lungcan.2019.05.027. Epub 2019 May 29. Lung Cancer. 2019. PMID: 31319991 Review.
Cited by
-
Existing Health Inequities in the Treatment of Advanced and Metastatic Cancers.Curr Oncol Rep. 2024 Dec;26(12):1553-1562. doi: 10.1007/s11912-024-01617-3. Epub 2024 Nov 4. Curr Oncol Rep. 2024. PMID: 39495424 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials