Individual glioblastoma cells harbor both proliferative and invasive capabilities during tumor progression
- PMID: 37335907
- PMCID: PMC10708941
- DOI: 10.1093/neuonc/noad109
Individual glioblastoma cells harbor both proliferative and invasive capabilities during tumor progression
Abstract
Background: Glioblastomas are characterized by aggressive and infiltrative growth, and by striking heterogeneity. The aim of this study was to investigate whether tumor cell proliferation and invasion are interrelated, or rather distinct features of different cell populations.
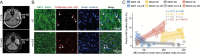
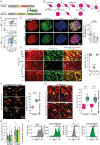
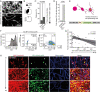
Methods: Tumor cell invasion and proliferation were longitudinally determined in real-time using 3D in vivo 2-photon laser scanning microscopy over weeks. Glioblastoma cells expressed fluorescent markers that permitted the identification of their mitotic history or their cycling versus non-cycling cell state.
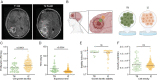
Results: Live reporter systems were established that allowed us to dynamically determine the invasive behavior, and previous or actual proliferation of distinct glioblastoma cells, in different tumor regions and disease stages over time. Particularly invasive tumor cells that migrated far away from the main tumor mass, when followed over weeks, had a history of marked proliferation and maintained their proliferative capacity during brain colonization. Infiltrating cells showed fewer connections to the multicellular tumor cell network, a typical feature of gliomas. Once tumor cells colonized a new brain region, their phenotype progressively transitioned into tumor microtube-rich, interconnected, slower-cycling glioblastoma cells. Analysis of resected human glioblastomas confirmed a higher proliferative potential of tumor cells from the invasion zone.
Conclusions: The detection of glioblastoma cells that harbor both particularly high proliferative and invasive capabilities during brain tumor progression provides valuable insights into the interrelatedness of proliferation and migration-2 central traits of malignancy in glioma. This contributes to our understanding of how the brain is efficiently colonized in this disease.
Keywords: cancer neuroscience; glioblastoma; migration; proliferation; tumor microtubes (TMs).
© The Author(s) 2023. Published by Oxford University Press on behalf of the Society for Neuro-Oncology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Conflict of interest statement
F.W., E.J., M.O. and W.W. report the patent (WO2017020982A1) “‘Agents for use in the treatment of glioma.’” F.W. is co-founder of DC Europa Ltd (a company trading under the name Divide and Conquer) that is developing new medicines for the treatment of glioma. Divide and Conquer also provides research funding to F.W.’s lab under a research collaboration agreement.
Figures


Comment in
-
Glioblastoma cell invasion: Go? Grow? Yes.Neuro Oncol. 2023 Dec 8;25(12):2163-2164. doi: 10.1093/neuonc/noad178. Neuro Oncol. 2023. PMID: 37739005 Free PMC article. No abstract available.
References
-
- Venkataramani V, Yang Y, Schubert MC, et al. Glioblastoma hijacks neuronal mechanisms for brain invasion. Cell. 2022;185(16):2899–2917. - PubMed
-
- Giese A, Bjerkvig R, Berens ME, Westphal M.. Cost of migration: Invasion of malignant gliomas and implications for treatment. J Clin Oncol. 2003;21(8):1624–1636. - PubMed
-
- Sahm F, Capper D, Jeibmann A, et al. Addressing diffuse glioma as a systemic brain disease with single-cell analysis. Arch Neurol. 2012;69(4):523–526. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

