Pharmacogenetics and pharmacokinetics of tamoxifen in a Zimbabwean breast cancer cohort
- PMID: 37337448
- PMCID: PMC10529681
- DOI: 10.1111/bcp.15827
Pharmacogenetics and pharmacokinetics of tamoxifen in a Zimbabwean breast cancer cohort
Abstract
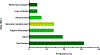
Tamoxifen is the most used hormonal therapy for oestrogen receptor-positive breast cancer. CYP2D6 is the main enzyme in the metabolic pathway of tamoxifen to endoxifen. Variations in endoxifen plasma concentrations are associated with CYP2D6 polymorphisms. This study aimed to determine the association between the CYP2D6 polymorphisms and endoxifen plasma concentrations in a cohort of Zimbabwean breast cancer patients (n = 40). TaqMan genotyping and copy number assays were done to determine CYP2D6 genotypes. Tamoxifen and metabolites were quantitated using LC-MS/MS. The population had high frequencies of the CYP2D6 reduced function alleles, *17 (15%) and *29 (18%). The median endoxifen concentration was 4.78 ng/mL, and in 55% of the patients, mostly intermediate metabolizers were below the endoxifen therapeutic threshold of 5.97 ng/mL. The CYP2D6 phenotypes and activity scores were significantly associated with endoxifen plasma concentrations (P = 0.0151) and with endoxifen to N-desmethyl-tamoxifen ratios (P = 0.0006).
Keywords: CYP2D6; breast cancer; pharmacogenetics; pharmacokinetics; tamoxifen.
© 2023 British Pharmacological Society.
Conflict of interest statement
Conflicts of Interest:
The authors declare no conflict of interest
Figures
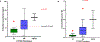
Similar articles
-
CYP2D6 polymorphisms and endoxifen concentration in Chinese patients with breast cancer.BMC Cancer. 2025 Mar 6;25(1):410. doi: 10.1186/s12885-025-13791-z. BMC Cancer. 2025. PMID: 40050768 Free PMC article.
-
Tamoxifen Pharmacogenetics and Metabolism: Results From the Prospective CYPTAM Study.J Clin Oncol. 2019 Mar 10;37(8):636-646. doi: 10.1200/JCO.18.00307. Epub 2019 Jan 24. J Clin Oncol. 2019. PMID: 30676859
-
Limited predictive value of achieving beneficial plasma (Z)-endoxifen threshold level by CYP2D6 genotyping in tamoxifen-treated Polish women with breast cancer.BMC Cancer. 2015 Aug 1;15:570. doi: 10.1186/s12885-015-1575-4. BMC Cancer. 2015. PMID: 26232141 Free PMC article.
-
CYP2D6 and tamoxifen: DNA matters in breast cancer.Nat Rev Cancer. 2009 Aug;9(8):576-86. doi: 10.1038/nrc2683. Nat Rev Cancer. 2009. PMID: 19629072 Review.
-
Clinical pharmacokinetics and pharmacogenetics of tamoxifen and endoxifen.Expert Rev Clin Pharmacol. 2019 Jun;12(6):523-536. doi: 10.1080/17512433.2019.1610390. Epub 2019 Apr 30. Expert Rev Clin Pharmacol. 2019. PMID: 31008668 Review.
Cited by
-
CYP2D6 polymorphisms and endoxifen concentration in Chinese patients with breast cancer.BMC Cancer. 2025 Mar 6;25(1):410. doi: 10.1186/s12885-025-13791-z. BMC Cancer. 2025. PMID: 40050768 Free PMC article.
-
CYP2D6 Genotyping for Optimization of Tamoxifen Therapy in Indonesian Women with ER+ Breast Cancer.J Pers Med. 2025 Feb 28;15(3):93. doi: 10.3390/jpm15030093. J Pers Med. 2025. PMID: 40137409 Free PMC article.
-
Precision pharmacotherapy of atomoxetine in children with ADHD: how to ensure the right dose for the right person?Front Pharmacol. 2024 Oct 29;15:1484512. doi: 10.3389/fphar.2024.1484512. eCollection 2024. Front Pharmacol. 2024. PMID: 39534083 Free PMC article. Review.
References
-
- Collaboration GBoDC. Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life Years for 29 Cancer Groups From 2010 to 2019: A Systematic Analysis for the Global Burden of Disease Study 2019. JAMA Oncology. 2022;8(3):420–444. doi:10.1001/jamaoncol.2021.6987 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

