Intravenous BCG Vaccination of Diversity Outbred Mice Results in Moderately Enhanced Protection against Challenge with Mycobacterium tuberculosis Compared to Intradermal Vaccination
- PMID: 37338410
- PMCID: PMC10353467
- DOI: 10.1128/iai.00168-23
Intravenous BCG Vaccination of Diversity Outbred Mice Results in Moderately Enhanced Protection against Challenge with Mycobacterium tuberculosis Compared to Intradermal Vaccination
Abstract
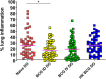
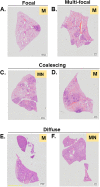
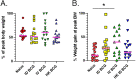
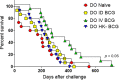
Tuberculosis is still the leading cause of death globally from any infectious disease, despite the widespread use of the live attenuated vaccine Bacille Calmette Guerin (BCG). While BCG has some efficacy against disseminated TB disease in children, protection wanes into adulthood resulting in over 1.8 million TB deaths per year. This has led to efforts to develop novel vaccine candidates that either replace or boost BCG, as well as to test novel delivery mechanisms to enhance BCG's efficacy. Traditional BCG vaccination is performed as an intradermal (ID) injection but delivering BCG by an alternate route may enhance the depth and breadth of protection. Previously, we demonstrated that phenotypically and genotypically disparate Diversity Outbred (DO) mice have heterogenous responses to M. tuberculosis challenge following intradermal BCG vaccination. Here, we utilize DO mice to examine BCG-induced protection when BCG is delivered systemically via intravenous (IV) administration. We find that DO mice vaccinated with IV BCG had a greater distribution of BCG throughout their organs compared to ID-vaccinated animals. However, compared to ID-vaccinated mice, M. tuberculosis burdens in lungs and spleens were not significantly reduced in animals vaccinated with BCG IV, nor was lung inflammation significantly altered. Nonetheless, DO mice that received BCG IV had increased survival over those vaccinated by the traditional ID route. Thus, our results suggest that delivering BCG by the alternate IV route enhances protection as detected in this diverse small animal model.
Keywords: BCG; Mycobacterium tuberculosis; diversity outbred; intravenous; mycobacterium; route; tuberculosis; vaccine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Multiple genetic loci influence vaccine-induced protection against Mycobacterium tuberculosis in genetically diverse mice.PLoS Pathog. 2024 Mar 7;20(3):e1012069. doi: 10.1371/journal.ppat.1012069. eCollection 2024 Mar. PLoS Pathog. 2024. PMID: 38452145 Free PMC article.
-
Intravenous BCG vaccination in non-human primates induces superior serum antibody titers with enhanced avidity and opsonizing capacity compared to the intradermal route.Vaccine. 2024 Dec 2;42(26):126444. doi: 10.1016/j.vaccine.2024.126444. Epub 2024 Nov 10. Vaccine. 2024. PMID: 39522337 Free PMC article.
-
The Diversity Outbred Mouse Population Is an Improved Animal Model of Vaccination against Tuberculosis That Reflects Heterogeneity of Protection.mSphere. 2020 Apr 15;5(2):e00097-20. doi: 10.1128/mSphere.00097-20. mSphere. 2020. PMID: 32295871 Free PMC article.
-
Protection against tuberculosis by Bacillus Calmette-Guérin (BCG) vaccination: A historical perspective.Med. 2022 Jan 14;3(1):6-24. doi: 10.1016/j.medj.2021.11.006. Epub 2021 Dec 31. Med. 2022. PMID: 35590145 Review.
-
[Novel vaccines against M. tuberculosis].Kekkaku. 2006 Dec;81(12):745-51. Kekkaku. 2006. PMID: 17240920 Review. Japanese.
Cited by
-
A review of the efficacy of clinical tuberculosis vaccine candidates in mouse models.Front Immunol. 2025 May 29;16:1609136. doi: 10.3389/fimmu.2025.1609136. eCollection 2025. Front Immunol. 2025. PMID: 40510368 Free PMC article. Review.
-
Host and pathogen genetic diversity shape vaccine-mediated protection to Mycobacterium tuberculosis.Front Immunol. 2024 Jun 28;15:1427846. doi: 10.3389/fimmu.2024.1427846. eCollection 2024. Front Immunol. 2024. PMID: 39007152 Free PMC article.
-
Mt10 Vaccine Protects Diversity Outbred Mice from CVB3 Infection by Producing Virus-Specific Neutralizing Antibodies and Diverse Antibody Isotypes.Vaccines (Basel). 2024 Mar 4;12(3):266. doi: 10.3390/vaccines12030266. Vaccines (Basel). 2024. PMID: 38543901 Free PMC article.
-
Deconvoluting the interplay of innate and adaptive immunity in BCG-induced nonspecific and TB-specific host resistance.J Exp Med. 2025 Apr 7;222(4):e20240496. doi: 10.1084/jem.20240496. Epub 2025 Mar 18. J Exp Med. 2025. PMID: 40100096 Review.
-
Recombinant BCGΔBCG1419c protects outbred mice against M. tuberculosis challenge.Vaccine. 2025 Aug 13;61:127347. doi: 10.1016/j.vaccine.2025.127347. Epub 2025 Jun 3. Vaccine. 2025. PMID: 40466484
References
-
- World Health Organization. 2022. Global tuberculosis report 2022.
-
- Nemes E, Geldenhuys H, Rozot V, Rutkowski KT, Ratangee F, Bilek N, Mabwe S, Makhethe L, Erasmus M, Toefy A, Mulenga H, Hanekom WA, Self SG, Bekker LG, Ryall R, Gurunathan S, DiazGranados CA, Andersen P, Kromann I, Evans T, Ellis RD, Landry B, Hokey DA, Hopkins R, Ginsberg AM, Scriba TJ, Hatherill M, Team C–S . 2018. Prevention of M. tuberculosis infection with H4:IC31 vaccine or BCG revaccination. N Engl J Med 379:138–149. doi:10.1056/NEJMoa1714021. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

