Ce(III) and La(III) ions adsorption using Amberlite XAD-7 resin impregnated with DEHPA extractant: response surface methodology, isotherm and kinetic study
- PMID: 37340031
- PMCID: PMC10282099
- DOI: 10.1038/s41598-023-37136-7
Ce(III) and La(III) ions adsorption using Amberlite XAD-7 resin impregnated with DEHPA extractant: response surface methodology, isotherm and kinetic study
Abstract
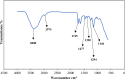
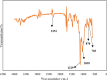
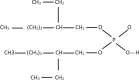
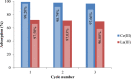
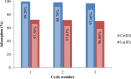
In this paper, the removal efficiency of Cerium (Ce(ΙΙΙ)) and lanthanum (La(ΙΙΙ)) ions from aqueous solution using Amberlite XAD-7 resin impregnated with DEHPA(XAD7-DEHPA) was studied in the batch system. The adsorbent ( XAD7-DEHPA) was characterized by SEM-EDX, FTIR and BET analysis Techniques. The response surface methodology based on the central composite design was applied to model and optimize the removal process and evaluate operating parameters like adsorbent dose (0.05-0.065), initial pH (2-6) and temperature (15-55). Variance analysis showed that the adsorbent dose, pH and temperature were the most effective parameters in the adsorption of Ce(ΙIΙ)and La(IΙI) respectively. The results showed that the optimum adsorption condition was achieved at pH = 6, the optimum amount of absorbent and the equilibrium time equal to 0.6 gr and 180 min, respectively. According to the results, the adsorption percentage of Ce(ΙIΙ) and La(ΙΙΙ) ions onto the aforementioned resin were 99.99% and 78.76% respectively. Langmuir, Freundlich, Temkin and sips isotherm models were applied to describe the equilibrium data. From the results, Langmuir isotherm (R2 (Ce) = 0.999, R2 (La) = 0.998) was found to better correlate the experimental rate data. The maximum adsorption capacity of the adsorbent ( XAD7-DEHPA) for both Ce(IΙI) and La(III) was found to be 8.28 and 5.52 mg g-1 respectively. The kinetic data were fitted to pseudo-first-order, pseudo-second-order and Intra particle diffusion models. Based on the results, the pseudo-first-order model and Intra particle diffusion model described the experimental data as well. In general, the results showed that ( XAD7-DEHPA) resin is an effective adsorbent for the removal of Ce(IΙI) and La(III) ions from aqueous solutions due to its high ability to selectively remove these metals as well as its reusability.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures











References
-
- Yuan H, et al. Extraction and back-extraction behaviors of 14 rare earth elements from sulfuric acid medium by TODGA. J. Rare Earths. 2018;36:642–647. doi: 10.1016/j.jre.2018.01.011. - DOI
-
- Jiang Z, et al. Adsorption of sulfonamides on polyamide microplastics in an aqueous solution: behavior, structural effects, and its mechanism. Chem. Eng. J. 2023;454:140452. doi: 10.1016/j.cej.2022.140452. - DOI
-
- Anastopoulos I, Bhatnagar A, Lima EC. Adsorption of rare earth metals: A review of recent literature. J. Mol. Liq. 2016;221:954–962. doi: 10.1016/j.molliq.2016.06.076. - DOI
-
- Wu K, et al. Adsorption of aqueous Cu (II) and Ag (I) by silica anchored Schiff base decorated polyamidoamine dendrimers: Behavior and mechanism. Chin. Chem. Lett. 2022;33:2721–2725. doi: 10.1016/j.cclet.2021.08.126. - DOI
LinkOut - more resources
Full Text Sources

