Objective wearable measures and subjective questionnaires for predicting response to neurostimulation in people with chronic pain
- PMID: 37340467
- PMCID: PMC10283222
- DOI: 10.1186/s42234-023-00115-4
Objective wearable measures and subjective questionnaires for predicting response to neurostimulation in people with chronic pain
Abstract
Background: Neurostimulation is an effective therapy for treating and management of refractory chronic pain. However, the complex nature of pain and infrequent in-clinic visits, determining subject's long-term response to the therapy remains difficult. Frequent measurement of pain in this population can help with early diagnosis, disease progression monitoring, and evaluating long-term therapeutic efficacy. This paper compares the utilization of the common subjective patient-reported outcomes with objective measures captured through a wearable device for predicting the response to neurostimulation therapy.
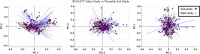
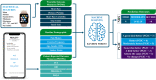
Method: Data is from the ongoing international prospective post-market REALITY clinical study, which collects long-term patient-reported outcomes from 557 subjects implanted by Spinal Cord Stimulator (SCS) or Dorsal Root Ganglia (DRG) neurostimulators. The REALITY sub-study was designed for collecting additional wearables data on a subset of 20 participants implanted with SCS devices for up to six months post implantation. We first implemented a combination of dimensionality reduction algorithms and correlation analyses to explore the mathematical relationships between objective wearable data and subjective patient-reported outcomes. We then developed machine learning models to predict therapy outcome based on the subject's response to the numerical rating scale (NRS) or patient global impression of change (PGIC).
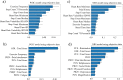
Results: Principal component analysis showed that psychological aspects of pain were associated with heart rate variability, while movement-related measures were strongly associated with patient-reported outcomes related to physical function and social role participation. Our machine learning models using objective wearable data predicted PGIC and NRS outcomes with high accuracy without subjective data. The prediction accuracy was higher for PGIC compared with the NRS using subjective-only measures primarily driven by the patient satisfaction feature. Similarly, the PGIC questions reflect an overall change since the study onset and could be a better predictor of long-term neurostimulation therapy outcome.
Conclusions: The significance of this study is to introduce a novel use of wearable data collected from a subset of patients to capture multi-dimensional aspects of pain and compare the prediction power with the subjective data from a larger data set. The discovery of pain digital biomarkers could result in a better understanding of the patient's response to therapy and their general well-being.
Keywords: Chronic pain therapy; Digital biomarkers for pain; Dimensionality reduction; Machine learning; PGIC; Patient-reported outcomes; Predictive modeling; Questionnaire burden; Spinal cord stimulation; Wearable technology.
© 2023. The Author(s).
Conflict of interest statement
SD, AK, and MM are employees of Abbott.
Figures


References
-
- Abdi H, Williams LJ. Principal component analysis. Wiley Interdiscip Rev Comput Stat. 2010;2(4):433–459. doi: 10.1002/wics.101. - DOI
-
- Breiman L. Random Forests. Mach Learn. 2001;45(1):5–32. doi: 10.1023/A:1010933404324. - DOI
-
- Caramia C, Torricelli D, Schmid M, Munoz-Gonzalez A, Gonzalez-Vargas J, Grandas F, et al. IMU-based classification of Parkinson's disease from gait: A sensitivity analysis on sensor location and feature selection. IEEE J Biomed Health Inform. 2018;22(6):1765–1774. doi: 10.1109/JBHI.2018.2865218. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
