Synthesis of New Chromene Derivatives Targeting Triple-Negative Breast Cancer Cells
- PMID: 37345018
- PMCID: PMC10216081
- DOI: 10.3390/cancers15102682
Synthesis of New Chromene Derivatives Targeting Triple-Negative Breast Cancer Cells
Abstract
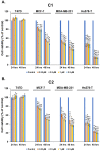
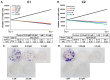
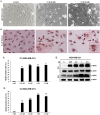
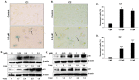
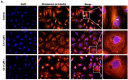
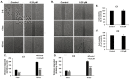
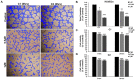
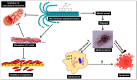
Breast cancer continues to be the leading cause of cancer-related deaths among women worldwide. The most aggressive type of breast cancer is triple-negative breast cancer (TNBC). Indeed, not only does TNBC not respond well to several chemotherapeutic agents, but it also frequently develops resistance to various anti-cancer drugs, including taxane mitotic inhibitors. This necessitates the search for newer, more efficacious drugs. In this study, we synthesized two novel chromene derivatives (C1 and C2) and tested their efficacy against a battery of luminal type A and TNBC cell lines. Our results show that C1 and C2 significantly and specifically inhibited TNBC cell viability but had no effect on the luminal A cell type. In addition, these novel compounds induced mitotic arrest, cell multinucleation leading to senescence, and apoptotic cell death through the activation of the extrinsic pathway. We also showed that the underlying mechanisms for these actions of C1 and C2 involved inhibition of microtubule polymerization and disruption of the F-actin cytoskeleton. Furthermore, both compounds significantly attenuated migration of TNBC cells and inhibited angiogenesis in vitro. Finally, we performed an in silico analysis, which revealed that these novel variants bind to the colchicine binding site in β-tubulin. Taken together, our data highlight the potential chemotherapeutic properties of two novel chromene compounds against TNBC.
Keywords: apoptosis; chromenes; microtubules; mitotic slippage; multinucleation; senescence.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





References
-
- World Health Organization Breast Cancer. [(accessed on 8 February 2023)]. Available online: https://www.who.int/news-room/fact-sheets/detail/breast-cancer.
-
- Orrantia-Borunda E., Anchondo-Nuñez P., Acuña-Aguilar L.E., Gómez-Valles F.O., Ramírez-Valdespino C.A. Subtypes of Breast Cancer. In: Mayrovitz H.N., editor. Breast Cancer. Exon Publications; Brisbane, Australia: 2022. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

