Clinical and Histopathological Features of an Italian Monocentric Series of Primary Small Bowel T-Cell Lymphomas
- PMID: 37345080
- PMCID: PMC10216175
- DOI: 10.3390/cancers15102743
Clinical and Histopathological Features of an Italian Monocentric Series of Primary Small Bowel T-Cell Lymphomas
Abstract
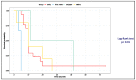
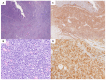
The gastrointestinal (GI) tract is the most common extranodal site of occurrence of non-Hodgkin lymphomas. Most GI lymphomas are of B-cell lineage, while T-cell lymphomas are less frequent. The aim of our retrospective study was to depict the clinical-pathological profile of a series of patients affected by intestinal T-cell lymphomas (ITCL) and possibly define hallmarks of these neoplasms. A total of 28 patients were included: 17 enteropathy-associated T-cell lymphomas (EATL), 5 monomorphic epitheliotropic T-cell lymphomas (MEITL), 3 indolent T-cell lymphoproliferative disorders of the gastrointestinal tract (ITCLDGT), and 3 intestinal T-cell lymphomas not otherwise specified (ITCL-NOS). Celiac disease (CD) was diagnosed in around 70% of cases. Diagnosis of EATL showed a significant correlation with CD30 expression, whereas MEITL with angiotropism and CD56 positivity. ITCLDGT cases showed plasma cells infiltration. Peripheral lymphocytosis, the absence of a previous diagnosis of CD, an advanced Lugano clinical stage, and the histological subtype ITCL-NOS were significantly associated with worse survival at multivariate analysis. Our findings about the epidemiological, clinical, and histopathological features of ITCL were in line with the current knowledge. Reliable prognostic tools for these neoplasms are still lacking but according to our results lymphocytosis, diagnosis of CD, Lugano clinical stage, and histological subtype should be considered for patient stratification.
Keywords: EATL; ITCL; ITCLDGT; MEITL; celiac disease; lymphoproliferative disorders; small bowel lymphoma.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
LinkOut - more resources
Full Text Sources
Research Materials

