Methotrexate vs expectant management for treatment of tubal ectopic pregnancy: An individual participant data meta-analysis
- PMID: 37345445
- PMCID: PMC10407021
- DOI: 10.1111/aogs.14617
Methotrexate vs expectant management for treatment of tubal ectopic pregnancy: An individual participant data meta-analysis
Abstract
Introduction: Ectopic pregnancy is an important health condition which affects up to 1 in 100 women. Women who present with mild symptoms and low serum human chorionic gonadotrophin (hCG) are often treated with methotrexate (MTX), but expectant management with close monitoring is a feasible alternative. Studies comparing the two treatments have not shown a statistically significant difference in uneventful resolution of ectopic pregnancy, but these studies were too small to define whether certain subgroups could benefit more from either treatment.
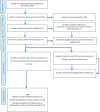
Material and methods: We performed a systematic review and individual participant data meta-analysis (IPD-MA) of randomized controlled trials comparing systemic MTX and expectant management in women with tubal ectopic pregnancy and low hCG (<2000 IU/L). A one-stage IPD-MA was performed to assess overall treatment effects of MTX and expectant management to generate a pooled intervention effect. Subgroup analyses and exploratory multivariable analyses were undertaken according to baseline serum hCG and progesterone levels. Primary outcome was treatment success, defined as resolution of clinical symptoms and decline in level of serum hCG to <20 IU/L, or a negative urine pregnancy test by the initial intervention strategy, without any additional treatment. Secondary outcomes were need for blood transfusion, surgical intervention, additional MTX side-effects and hCG resolution times.
Trial registration number: PROSPERO: CRD42021214093.
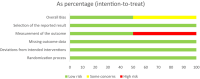
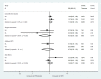
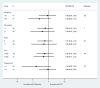
Results: 1547 studies reviewed and 821 remained after duplicates removed. Five studies screened for eligibility and three IPD requested. Two randomized controlled trials supplied IPD, leading to 153 participants for analysis. Treatment success rate was 65/82 (79.3%) after MTX and 48/70 (68.6%) after expectant management (IPD risk ratio [RR] 1.16, 95% confidence interval [CI] 0.95-1.40). Surgical intervention rates were not significantly different: 8/82 (9.8%) vs 13/70 (18.6%) (RR 0.65, 95% CI 0.23-1.14). Mean time to success was 19.7 days (95% CI 17.4-22.3) after MTX and 21.2 days (95% CI 17.8-25.2) after expectant management (P = 0.25). MTX specific side-effects were reported in 33 MTX compared to four in the expectant group.
Conclusions: Our IPD-MA showed no statistically significant difference in treatment efficacy between MTX and expectant management in women with tubal ectopic pregnancy with low hCG. Initial expectant management could be the preferred strategy due to fewer side-effects.
Keywords: expectant management; medical treatment; methotrexate; pregnancy ectopic; pregnancy tubal.
© 2023 The Authors. Acta Obstetricia et Gynecologica Scandinavica published by John Wiley & Sons Ltd on behalf of Nordic Federation of Societies of Obstetrics and Gynecology (NFOG).
Conflict of interest statement
BWM is supported by a NHMRC Investigatorgrant (GNT1176437). BWM reports consultancy for ObsEva and Merck and travel support from Merck. The other authors do not have any conflict of interests to declare.
Figures



References
-
- O'Herlihy C. Centre for Maternal and Child Enquiries (CMACE). Deaths in early pregnancy. Saving mother's lives: reviewing maternal deaths to make motherhood safer: 2006‐2008. The Eighth Report of the Confidential Enquiries into Maternal Deaths in the United Kingdom. BJOG. 2011;118(Suppl.1):81‐84. - PubMed
-
- Dooley WM, Chaggar P, De Braud LV, Bottomley C, Jauniaux E, Jurkovic D. Effect of morphological type of extrauterine ectopic pregnancy on accuracy of preoperative ultrasound diagnosis. Ultrasound Obstet Gynecol. 2019;54:538‐544. - PubMed
-
- Rodi IA, Sauer MV, Gorrill MJ, et al. The medical treatment of unruptured ectopic pregnancy with methotrexate and citrovorum rescue: preliminary experience. Fertil Steril. 1986;46:811‐813. - PubMed
-
- Sauer MV, Gorrill MJ, Rodi IA, et al. Nonsurgical management of unruptured ectopic pregnancy: an extended clinical trial. Fertil Steril. 1987;48:752‐755. - PubMed
-
- Cacciatore B, Korhonen J, Stenman UH, Ylostalo P. Transvaginal sonography and serum hCG in monitoring of presumed ectopic pregnancies selected for expectant management. Ultrasound Obstet Gynecol. 1995;5:297‐300. - PubMed

