Disruption of the sialic acid/Siglec-9 axis improves antibody-mediated neutrophil cytotoxicity towards tumor cells
- PMID: 37346044
- PMCID: PMC10279866
- DOI: 10.3389/fimmu.2023.1178817
Disruption of the sialic acid/Siglec-9 axis improves antibody-mediated neutrophil cytotoxicity towards tumor cells
Abstract
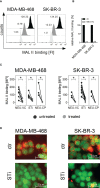
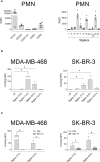
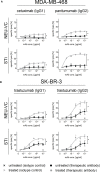
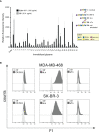
Upregulation of surface expressed sialoglycans on tumor cells is one of the mechanisms which promote tumor growth and progression. Specifically, the interactions of sialic acids with sialic acid-binding immunoglobulin-like lectins (Siglecs) on lymphoid or myeloid cells transmit inhibitory signals and lead to suppression of anti-tumor responses. Here, we show that neutrophils express among others Siglec-9, and that EGFR and HER2 positive breast tumor cells express ligands for Siglec-9. Treatment of tumor cells with neuraminidases or a sialyl transferase inhibitor significantly reduced binding of a soluble recombinant Siglec-9-Fc fusion protein, while EGFR and HER2 expression remained unchanged. Importantly, the cytotoxic activity of neutrophils driven by therapeutic EGFR or HER2 antibodies in vitro was increased by blocking the sialic acid/Siglec interaction, either by reducing tumor cell sialylation or by a Siglec-9 blocking antibody containing an effector silenced Fc domain. In vivo a short-term xenograft mouse model confirmed the improved therapeutic efficacy of EGFR antibodies against sialic acid depleted, by a sialyltransferase inhibitor, tumor cells compared to untreated cells. Our studies demonstrate that sialic acid/Siglec interactions between tumor cells and myeloid cells can impair antibody dependent tumor cell killing, and that Siglec-9 on polymorphonuclear cells (PMN) is critically involved. Considering that PMN are often a highly abundant cell population in the tumor microenvironment, Siglec-9 constitutes a promising target for myeloid checkpoint blockade to improve antibody-based tumor immunotherapy.
Keywords: ADCC - antibody dependent cellular cytotoxicity; Siglec-9; glycans; myeloid cells; neutrophils (PMNs); sialic acid; siglecs.
Copyright © 2023 Lustig, Chan, Jansen, Bräutigam, Kölling, Gehlert, Baumann, Mester, Foss, Andersen, Bastian, Sondermann, Peipp, Burger, Leusen and Valerius.
Conflict of interest statement
MB and PS are employed by Tacalyx GmbH. JHWL is scientific founder and shareholder of TigaTx. All other authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Gagneux P, Hennet T, Varki A. Biological functions of glycans. In: Varki A, Cummings RD, Esko JD, Stanley P, Hart GW, Aebi M, et al., editors. Essentials of glycobiology. NY: Cold Spring Harbor; (2022). p. 79–92.
-
- Varki A, Kannagi R, Toole B, Stanley P. Glycosylation changes in cancer. In: Varki A, Cummings RD, Esko JD, Stanley P, Hart GW, et al., editors. Essentials of glycobiology. NY: Cold Spring Harbor; (2015). p. 597–609.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

