Experience alters hippocampal and cortical network communication via a KIBRA-dependent mechanism
- PMID: 37347662
- PMCID: PMC10592482
- DOI: 10.1016/j.celrep.2023.112662
Experience alters hippocampal and cortical network communication via a KIBRA-dependent mechanism
Abstract
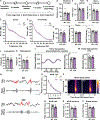
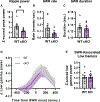
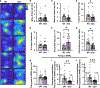
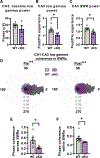
Synaptic plasticity is hypothesized to underlie "replay" of salient experience during hippocampal sharp-wave/ripple (SWR)-based ensemble activity and to facilitate systems-level memory consolidation coordinated by SWRs and cortical sleep spindles. It remains unclear how molecular changes at synapses contribute to experience-induced modification of network function. The synaptic protein KIBRA regulates plasticity and memory. To determine the impact of KIBRA-regulated plasticity on circuit dynamics, we recorded in vivo neural activity from wild-type (WT) mice and littermates lacking KIBRA and examined circuit function before, during, and after novel experience. In WT mice, experience altered population activity and oscillatory dynamics in a manner consistent with incorporation of new information content in replay and enhanced hippocampal-cortical communication. While baseline SWR features were normal in KIBRA conditional knockout (cKO) mice, experience-dependent alterations in SWRs were absent. Furthermore, intra-hippocampal and hippocampal-cortical communication during SWRs was disrupted following KIBRA deletion. These results indicate molecular mechanisms that underlie network-level adaptations to experience.
Keywords: CP: Neuroscience; KIBRA; anterior cingulate cortex; hippocampus; place cell; plasticity; sharp-wave ripple; sleep spindle.
Copyright © 2023 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures



References
-
- Tonegawa S, Tsien JZ, McHugh TJ, Huerta P, Blum KI, and Wilson MA (1996). Hippocampal CA1-region-restricted knockout of NMDAR1 gene disrupts synaptic plasticity, place fields, and spatial learning. Cold Spring Harbor Symp. Quant. Biol. 61, 225–238. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

