AMEERA-3: Randomized Phase II Study of Amcenestrant (Oral Selective Estrogen Receptor Degrader) Versus Standard Endocrine Monotherapy in Estrogen Receptor-Positive, Human Epidermal Growth Factor Receptor 2-Negative Advanced Breast Cancer
- PMID: 37348019
- PMCID: PMC10461947
- DOI: 10.1200/JCO.22.02746
AMEERA-3: Randomized Phase II Study of Amcenestrant (Oral Selective Estrogen Receptor Degrader) Versus Standard Endocrine Monotherapy in Estrogen Receptor-Positive, Human Epidermal Growth Factor Receptor 2-Negative Advanced Breast Cancer
Abstract
Purpose: Amcenestrant (oral selective estrogen receptor degrader) demonstrated promising safety and efficacy in earlier clinical studies for endocrine-resistant, estrogen receptor-positive/human epidermal growth factor receptor 2-negative (ER+/HER2-) advanced breast cancer (aBC).
Patients and methods: In AMEERA-3 (ClinicalTrials.gov identifier: NCT04059484), an open-label, worldwide phase II trial, patients with ER+/HER2- aBC who progressed in the (neo)adjuvant or advanced settings after not more than two previous lines of endocrine therapy (ET) were randomly assigned 1:1 to amcenestrant or single-agent endocrine treatment of physician's choice (TPC), stratified by the presence/absence of visceral metastases, previous/no treatment with cyclin-dependent kinase 4/6 inhibitor, and Eastern Cooperative Oncology Group performance status (0/1). The primary end point was progression-free survival (PFS) by independent central review, compared using a stratified log-rank test (one-sided type I error rate of 2.5%).
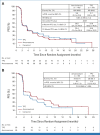
Results: Between October 22, 2019, and February 15, 2021, 290 patients were randomly assigned to amcenestrant (n = 143) or TPC (n = 147). PFS was numerically similar between amcenestrant and TPC (median PFS [mPFS], 3.6 v 3.7 months; stratified hazard ratio [HR], 1.051 [95% CI, 0.789 to 1.4]; one-sided P = .643). Among patients with baseline mutated ESR1; (n = 120 of 280), amcenestrant numerically prolonged PFS versus TPC (mPFS, 3.7 v 2.0 months; stratified HR, 0.9 [95% CI, 0.565 to 1.435]). Overall survival data were immature but numerically similar between groups (HR, 0.913; 95% CI, 0.595 to 1.403). In amcenestrant versus TPC groups, treatment-emergent adverse events (any grade) occurred in 82.5% versus 76.2% of patients and grade ≥3 events occurred in 21.7% versus 15.6%.
Conclusion: AMEERA-3 did not meet its primary objective of improved PFS with amcenestrant versus TPC although a numerical improvement in PFS was observed in patients with baseline ESR1 mutation. Efficacy and safety with amcenestrant were consistent with the standard of care for second-/third-line ET for ER+/HER2- aBC.
Conflict of interest statement
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
No other potential conflicts of interest were reported.
Figures


References
-
- Sung H, Ferlay J, Siegel RL, et al. : Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209-249, 2021 - PubMed
-
- National Cancer Institute Surveillance , Epidemiology, and End Results Program: Cancer stat facts: Female breast cancer subtypes. https://seer.cancer.gov/statfacts/html/breast-subtypes.html
-
- American Cancer Society : Breast Cancer Facts & Figures 2019-2020. Atlanta, GA, American Cancer Society, Inc, 2019
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

