A wireless patch for the monitoring of C-reactive protein in sweat
- PMID: 37349389
- PMCID: PMC10592261
- DOI: 10.1038/s41551-023-01059-5
A wireless patch for the monitoring of C-reactive protein in sweat
Abstract
The quantification of protein biomarkers in blood at picomolar-level sensitivity requires labour-intensive incubation and washing steps. Sensing proteins in sweat, which would allow for point-of-care monitoring, is hindered by the typically large interpersonal and intrapersonal variations in its composition. Here we report the design and performance of a wearable and wireless patch for the real-time electrochemical detection of the inflammatory biomarker C-reactive (CRP) protein in sweat. The device integrates iontophoretic sweat extraction, microfluidic channels for sweat sampling and for reagent routing and replacement, and a graphene-based sensor array for quantifying CRP (via an electrode functionalized with anti-CRP capture antibodies-conjugated gold nanoparticles), ionic strength, pH and temperature for the real-time calibration of the CRP sensor. In patients with chronic obstructive pulmonary disease, with active or past infections or who had heart failure, the elevated concentrations of CRP measured via the patch correlated well with the protein's levels in serum. Wearable biosensors for the real-time sensitive analysis of inflammatory proteins in sweat may facilitate the management of chronic diseases.
© 2023. The Author(s), under exclusive licence to Springer Nature Limited.
Conflict of interest statement
Competing interests
The authors declare no competing interests.
Figures
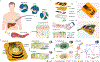




References
-
- The top 10 causes of death. https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death.
-
- Ashley NT, Weil ZM & Nelson RJ Inflammation: mechanisms, costs, and natural variation. Annu. Rev. Ecol. Evol. Syst 43, 385–406 (2012).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

