Synthesis of Quaternary and Tertiary Carbon-Substituted Arenes by Lewis Base Promoted Site-Selective Coupling with Allylic Nucleophiles
- PMID: 37350059
- PMCID: PMC10529890
- DOI: 10.1002/anie.202306277
Synthesis of Quaternary and Tertiary Carbon-Substituted Arenes by Lewis Base Promoted Site-Selective Coupling with Allylic Nucleophiles
Abstract
A practical method for the preparation of quaternary and tertiary allyl-substituted heteroarenes by site-selective couplings of heteroaryl nitriles and allylic nucleophiles is disclosed. Transformations utilize readily accessible stable reagents, proceed in the presence of a Lewis base activator, and undergo aryl-C(sp3 ) quaternary and tertiary carbon formation with high γ-selectivity (up to >98 : 2 γ : α).
Keywords: Allylation; Arene Coupling; Heterocycle; Organoboron; Quaternary Carbon.
© 2023 Wiley-VCH Verlag GmbH.
Conflict of interest statement
Conflict of Interest
The authors declare no conflict of interest.
Figures

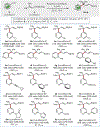

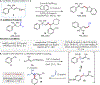
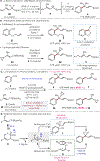
References
-
- For recent reviews on the enantioselective synthesis of quaternary carbon stereogenic centers, see: Das JP, Marek I, Chem. Commun. 2011, 47, 4593–4623. - PubMed
- Scott HK, Aggarwal VK, Chemistry – A European Journal 2011, 17, 13124–13132. - PubMed
- Minko Y, Marek I, Chem. Commun. 2014, 50, 12597–12611. - PubMed
- Marek I, Minko Y, Pasco M, Mejuch T, Gilboa N, Chechik H, Das JP, J. Am. Chem. Soc. 2014, 136, 2682–2694. - PubMed
- Quasdorf KW, Overman LE, Nature 2014, 516, 181–191. - PMC - PubMed
- Liu Y, Han S-J, Liu W-B, Stoltz BM, Acc. Chem. Res. 2015, 48, 740–751.Z - PMC - PubMed
- Zeng X-P, Cao Z-Y, Wang Y-H, Zhou F, J. Zhou, Chem. Rev. 2016, 116, 7330–7396. - PubMed
- Feng J, Holmes M, Krische MJ, Chem. Rev. 2017, 117, 12564–12580. - PMC - PubMed
- Süsse L, Stoltz BM, Chem. Rev. 2021, 121, 4084–4099. - PMC - PubMed
- Wright TB, Evans PA, Chem. Rev. 2021, 121, 9196–9242. - PubMed
- Yu X, Zhang T, Liu J, Li X, Synthesis 2021, 53, 4341–4352.
-
- For an example of an intramolecular Smiles rearrangement employing allylic nucleophiles see: Tetlow DJ, Hennecke U, Raftery J, Waring MJ, Clarke DS, Clayden J, Org. Lett. 2010, 12, 5442–5445. - PubMed
-
- Yamamoto Y, Takada S, Miyaura N, Iyama T, Tachikawa H, Organometallics 2009, 28, 152–160.
- Glasspoole BW, Ghozati K, Moir JW, Crudden CM, Chem. Commun. 2012, 48, 1230–1232. - PubMed
- Farmer JL, Hunter HN, Organ MG, J. Am. Chem. Soc. 2012, 134, 17470–17473. - PubMed
- Yang Y, Buchwald SL, J. Am. Chem. Soc. 2013, 135, 10642–10645. - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

