Fluctuations in Gut Microbiome Composition During Immune Checkpoint Inhibitor Therapy
- PMID: 37350807
- PMCID: PMC10284636
- DOI: 10.14740/wjon1587
Fluctuations in Gut Microbiome Composition During Immune Checkpoint Inhibitor Therapy
Abstract
Background: Immune checkpoint inhibitors (ICIs) such as programmed cell death protein-1 (PD-1) inhibitors or PD-1 ligand-1 (PD-L1) inhibitors have led to remarkable improvement in outcomes of non-small cell lung cancer (NSCLC). Unfortunately, the significant benefits of ICI therapy are frequently limited by resistance to treatment and adverse effects, and the predictive value of pre-treatment tumor tissue PD-L1 expression is limited. Development of less invasive biomarkers that could identify responders and non-responders in early on-treatment could markedly improve the treatment regimen. Accumulating evidence suggests that baseline gut microbiota profile is associated with response to PD-1/PD-L1 blockade therapy. However, change in the gut microbiome composition during PD-1/PD-L1 blockade therapy and its relation to response remain unclear.
Methods: Here, we analyzed pre- and on-treatment fecal samples from five NSCLC patients receiving anti-PD-1 immunotherapy, alone or in tandem with chemotherapy, and performed 16S rRNA sequencing.
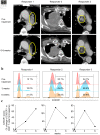
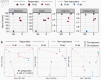
Results: The overall alpha diversity of the baseline gut microbiome was similar between three responders and two non-responders. While the gut microbiome composition remained stable overall during treatment (R2 = 0.145), responders showed significant changes in microbiome diversity between pre- and on-treatment samples during anti-PD-1 therapy compared to non-responders (P = 0.0274). Within the diverse microbiota, responders showed decreases in the abundance of genera Odoribacter, Gordonibacter, Candidatus Stoquefichus, Escherichia-Shigella, and Collinsella, and increase in abundance of Clostridium sensu stricto 1. In contrast, non-responders demonstrated on-treatment increases in genera Prevotella, Porphyromonas, Streptococcus, and Escherichia-Shigella, and decrease in abundance of Akkermansia.
Conclusions: This pilot study identified a substantial change in gut microbiome diversity between pre- and on-treatment samples in NSCLC patients responding to anti-PD-1 therapy compared to non-responders. Our findings highlight the potential utility of gut microbiota dynamics as a noninvasive biomarker to predict response to PD-1/PD-L1 blockade therapy for a wide variety of malignancies, which sets a path for future investigation in larger prospective studies.
Keywords: Biomarker; Gut microbiome; Immunotherapy; Non-small cell lung cancer; PD-1.
Copyright 2023, Sarkar et al.
Conflict of interest statement
The authors declare no potential conflicts of interest.
Figures

Similar articles
-
The impact of microbiota on PD-1/PD-L1 inhibitor therapy outcomes: A focus on solid tumors.Life Sci. 2022 Dec 1;310:121138. doi: 10.1016/j.lfs.2022.121138. Epub 2022 Oct 26. Life Sci. 2022. PMID: 36309224 Review.
-
Interaction of bacterial genera associated with therapeutic response to immune checkpoint PD-1 blockade in a United States cohort.Genome Med. 2022 Mar 29;14(1):35. doi: 10.1186/s13073-022-01037-7. Genome Med. 2022. PMID: 35346337 Free PMC article.
-
The gut microbiota modulates responses to anti-PD-1 and chemotherapy combination therapy and related adverse events in patients with advanced solid tumors.Front Oncol. 2022 Oct 25;12:887383. doi: 10.3389/fonc.2022.887383. eCollection 2022. Front Oncol. 2022. PMID: 36387171 Free PMC article.
-
The Diversity of Gut Microbiome is Associated With Favorable Responses to Anti-Programmed Death 1 Immunotherapy in Chinese Patients With NSCLC.J Thorac Oncol. 2019 Aug;14(8):1378-1389. doi: 10.1016/j.jtho.2019.04.007. Epub 2019 Apr 23. J Thorac Oncol. 2019. PMID: 31026576
-
The Gut Microbiome and Cancer Immunotherapy: Can We Use the Gut Microbiome as a Predictive Biomarker for Clinical Response in Cancer Immunotherapy?Cancers (Basel). 2021 Sep 27;13(19):4824. doi: 10.3390/cancers13194824. Cancers (Basel). 2021. PMID: 34638308 Free PMC article. Review.
Cited by
-
Gut microbes and immunotherapy for non-small cell lung cancer: a systematic review.Front Oncol. 2025 May 8;15:1518474. doi: 10.3389/fonc.2025.1518474. eCollection 2025. Front Oncol. 2025. PMID: 40406244 Free PMC article.
-
Fecal, duodenal, and tumor microbiota composition of esophageal carcinoma patients, a longitudinal prospective cohort.J Natl Cancer Inst. 2024 Nov 1;116(11):1834-1844. doi: 10.1093/jnci/djae153. J Natl Cancer Inst. 2024. PMID: 38924513 Free PMC article.
-
Gut microbiota and dietary intervention: affecting immunotherapy efficacy in non-small cell lung cancer.Front Immunol. 2024 Feb 1;15:1343450. doi: 10.3389/fimmu.2024.1343450. eCollection 2024. Front Immunol. 2024. PMID: 38361936 Free PMC article. Review.
-
Gut-lung axis: role of the gut microbiota in non-small cell lung cancer immunotherapy.Front Oncol. 2023 Nov 22;13:1257515. doi: 10.3389/fonc.2023.1257515. eCollection 2023. Front Oncol. 2023. PMID: 38074650 Free PMC article. Review.
References
-
- Herbst RS, Baas P, Kim DW, Felip E, Perez-Gracia JL, Han JY, Molina J. et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387(10027):1540–1550. doi: 10.1016/S0140-6736(15)01281-7. - DOI - PubMed
-
- Felip E, Altorki N, Zhou C, Csoszi T, Vynnychenko I, Goloborodko O, Luft A. et al. Adjuvant atezolizumab after adjuvant chemotherapy in resected stage IB-IIIA non-small-cell lung cancer (IMpower010): a randomised, multicentre, open-label, phase 3 trial. Lancet. 2021;398(10308):1344–1357. doi: 10.1016/S0140-6736(21)02098-5. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
