LNL-Carbazole Pincer Ligand: More than the Sum of Its Parts
- PMID: 37351619
- PMCID: PMC10347472
- DOI: 10.1021/acs.chemrev.3c00202
LNL-Carbazole Pincer Ligand: More than the Sum of Its Parts
Abstract
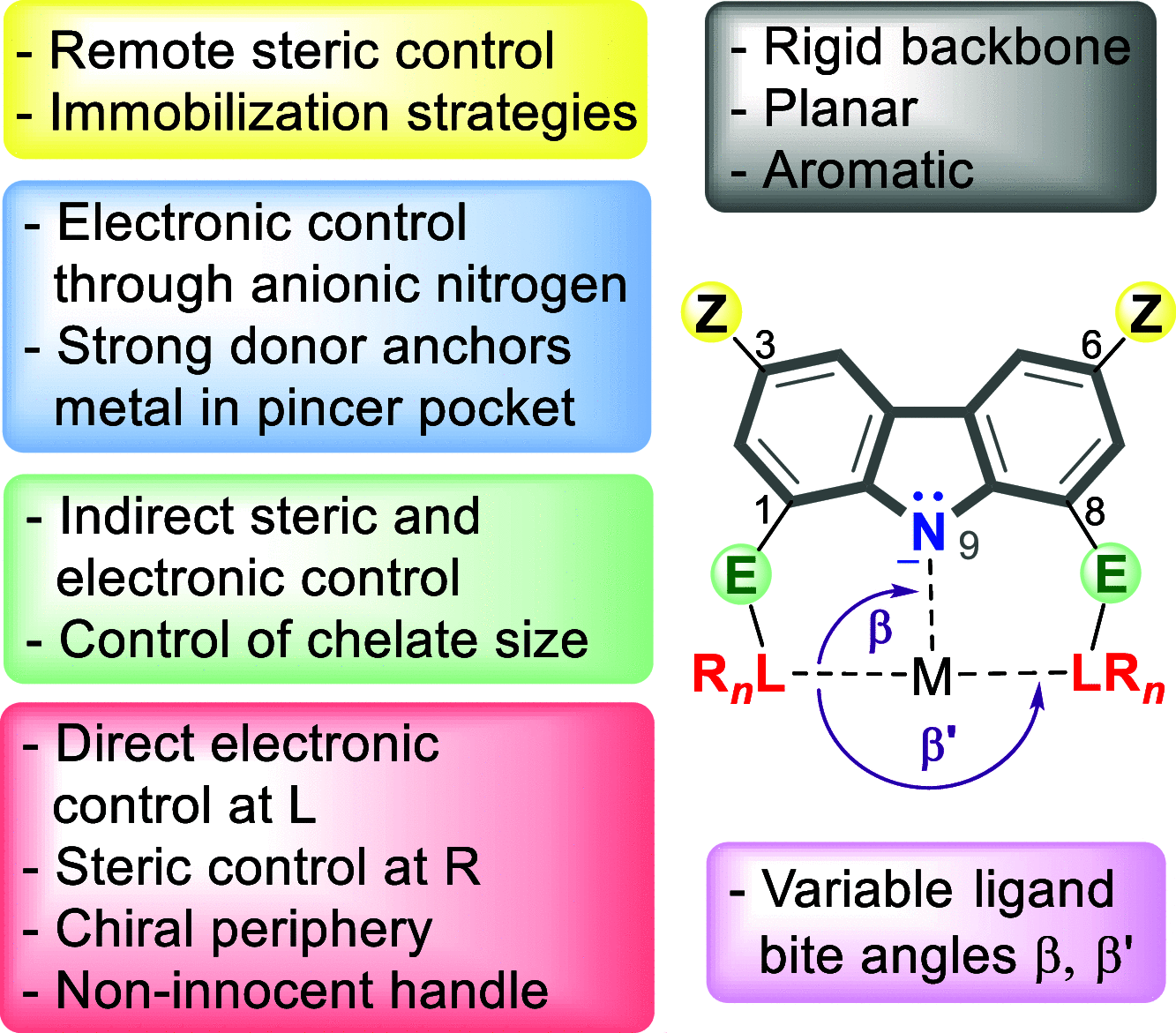
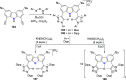
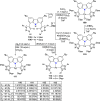
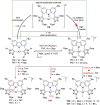
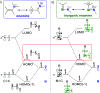
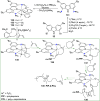
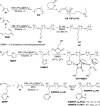
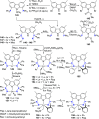
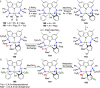
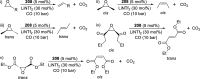
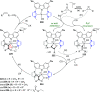
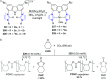
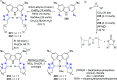
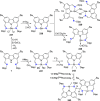
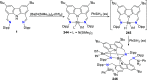
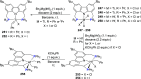
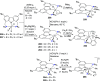
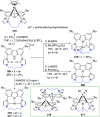
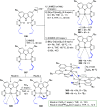
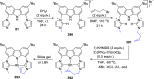
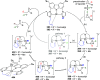
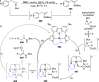
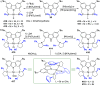
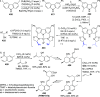
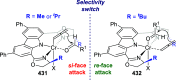
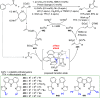
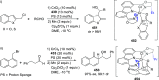
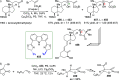
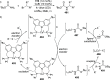
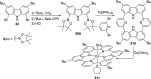
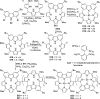
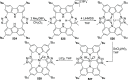
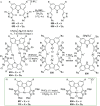
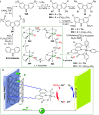
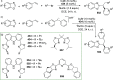
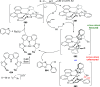
The utility of carbazole in photo-, electro-, and medicinal applications has ensured its widespread use also as the backbone in tridentate pincer ligands. In this review, the aim is to identify and illustrate the key features of the LNL-carbazolide binding to transition metal centers (with L = flanking donor moieties, e.g., C, N, P, and O-groups) in a systematic bottom-up progression to illustrate the marked benefits attainable from (i) the rigid aromatic carbazole scaffold (modulable in both the 1,8- and 3,6-positions), (ii) the significant electronic effect of central carbazole-amido binding to a metal, and the tunable sterics and electronics of both the (iii) flanking donor L-moieties and (iv) the wingtip R-groups on the L-donors, with their corresponding influence on metal coordination geometry, d-electron configuration, and resultant reactivity. Systematic implementation of the ligand design strategies not in isolation, but in a combinatorial approach, is showcased to demonstrate the potential for functional molecules that are not only modulable but also adaptable for wide-ranging applications (e.g., stereoselective (photo)catalysis, challenging small molecule activation, SET and redox applications, and even applications in chemotherapeutics) as an indication of future research efforts anticipated to stem from this versatile pincer assembly, not only for the transition metals but also for s-, p-, and f-block elements.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

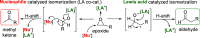


References
-
- Yin J.; Ma Y.; Li G.; Peng M.; Lin W. A Versatile Small-Molecule Fluorescence Scaffold: Carbazole Derivatives for Bioimaging. Coord. Chem. Rev. 2020, 412, 213257.10.1016/j.ccr.2020.213257. - DOI
-
- Manickam M.; Iqbal P.; Belloni M.; Kumar S.; Preece J. A. A Brief Review of Carbazole-Based Photorefractive Liquid Crystalline Materials. Isr. J. Chem. 2012, 52, 917–934. 10.1002/ijch.201200058. - DOI
-
- Mitra A. K. Sesquicentennial Birth Anniversary of Carbazole, a Multifaceted Wonder Molecule: A Revisit to Its Synthesis, Photophysical and Biological Studies. J. Iran. Chem. Soc. 2022, 19, 2075–2113. 10.1007/s13738-021-02444-0. - DOI
-
- Grigalevicius S. 3,6(2,7),9-Substituted Carbazoles as Electroactive Amorphous Materials for Optoelectronics. Synth. Met. 2006, 156, 1–12. 10.1016/j.synthmet.2005.10.004. - DOI
Publication types
LinkOut - more resources
Full Text Sources

