Rare subtypes of triple negative breast cancer: Current understanding and future directions
- PMID: 37353557
- PMCID: PMC10290136
- DOI: 10.1038/s41523-023-00554-x
Rare subtypes of triple negative breast cancer: Current understanding and future directions
Abstract
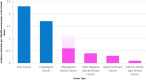
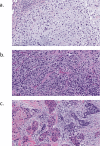
Rare subtypes of triple-negative breast cancers (TNBC) are a heterogenous group of tumors, comprising 5-10% of all TNBCs. Despite accounting for an absolute number of cases in aggregate approaching that of other less common, but well studied solid tumors, rare subtypes of triple-negative disease remain understudied. Low prevalence, diagnostic challenges and overlapping diagnoses have hindered consistent categorization of these breast cancers. Here we review epidemiology, histology and clinical and molecular characteristics of metaplastic, triple-negative lobular, apocrine, adenoid cystic, secretory and high-grade neuroendocrine TNBCs. Medullary pattern invasive ductal carcinoma no special type, which until recently was a considered a distinct subtype, is also discussed. With this background, we review how applying biological principals often applied to study TNBC no special type could improve our understanding of rare TNBCs. These could include the utilization of targeted molecular approaches or disease agnostic tools such as tumor mutational burden or germline mutation-directed treatments. Burgeoning data also suggest that pathologic response to neoadjuvant therapy and circulating tumor DNA have value in understanding rare subtypes of TNBC. Finally, we discuss a framework for advancing disease-specific knowledge in this space. While the conduct of randomized trials in rare TNBC subtypes has been challenging, re-envisioning trial design and technologic tools may offer new opportunities. These include embedding rare TNBC subtypes in umbrella studies of rare tumors, retrospective review of contemporary trials, prospective identification of patients with rare TNBC subtypes entering on clinical trials and querying big data for outcomes of patients with rare breast tumors.
© 2023. The Author(s).
Conflict of interest statement
A.T. reports Stock Ownership: Johnson & Johnson, Gilead Sciences, Bristol Myers Squibb, Pfizer, Doximity; Consulting or Advisory Role: BeyondSpring Pharmaceuticals, Lilly, Genentech, AstraZeneca; Research Funding: Sanofi (to the institution); Royalties UpToDate Royalties; J.S.R.-F. reports personal/consultancy fees from Goldman Sachs, Bain Capital, REPARE Therapeutics, Paige.AI and Personalis, membership of the scientific advisory boards of VolitionRx, REPARE Therapeutics, Paige.AI and Personalis, membership of the Board of Directors of Grupo Oncoclinicas, and ad hoc membership of the scientific advisory boards of Roche Tissue Diagnostics, Daiichi Sankyo, Merck, and AstraZeneca, outside the scope of this work. C.E.G. reports personal fees for advisory board membership from Exact Sciences; personal fees for consulting from Athenex; a role as Co-Chair of steering committees at Abbvie, Daiichi Sankyo and Genentech/Roche (non-financial interest); a role as coordinating PI for Genentech/Roche (non-financial interest); a non-renumerated role as Co-Chair of an executive committee and a steering committee at AstraZeneca; and a non-renumerated advisory role for AstraZeneca, Daiichi Sankyo, Genentech/Roche and Seagen. H.Y.W. reports Advisory Role for AstraZeneca. Additionally, author Jorge Reis-Filho is a Deputy Editor-in-Chief of
Figures







References
-
- DeSantis CE, Kramer JL, Jemal A. The burden of rare cancers in the United States. CA: A Cancer J. Clin. 2017;67:261–272. - PubMed
-
- WHO Classification of Tumors: Breast Tumours. 5 edn, Vol. 2 (International Agency for Research on Cancer, 2019).
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

