Non-paretic leg movements can facilitate cortical drive to the paretic leg in individuals post stroke with severe motor impairment: Implications for motor priming
- PMID: 37354080
- PMCID: PMC10530620
- DOI: 10.1111/ejn.16069
Non-paretic leg movements can facilitate cortical drive to the paretic leg in individuals post stroke with severe motor impairment: Implications for motor priming
Abstract
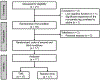
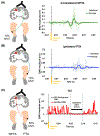
Cross-education, a phenomenon where unilateral strength (or skill) training enhances strength (or skill) in the contralateral untrained limb, has been well studied in able-bodied individuals. Cross-education effect accompanies bilateral changes of corticomotor activity in the motor cortex (M1). Recent reports demonstrated greater cross-education effect in stroke survivors compared to healthy individuals, however, corticomotor responses to cross-education in stroke remains unclear. This study aimed to determine the effects of non-paretic leg movements on corticomotor excitability (CME) and reaction time of the paretic leg in severely impaired stroke survivors. Seventeen post stroke individuals with severe leg motor impairment (Fugl-Meyer lower extremity score less than 21 and absence of motor evoked potential in the paretic leg) performed three 20-min motor trainings using their non-paretic ankle: skill (targeted dynamic movements), strength (isometric resistance) and sham (sub-threshold electrical nerve stimulation). During training, verbal instructions were given to the participants to limit their movement to the non-paretic leg and this was confirmed with visual observation of the paretic leg. Transcranial magnetic stimulation measured CME of the contralateral pathways from the non-lesioned M1 to the non-paretic tibialis anterior (TA) muscle, ipsilateral pathways to the paretic TA and transcallosal inhibition (TCI) from the non-lesioned to lesioned M1. Paretic ankle reaction time was measured using a reaction time paradigm. All outcomes were measured before, immediately post, 30-min post and 60-min post priming. CME of the non-paretic TA increased after skill (.08 ± .10 mV) and strength (.06 ± .05 mV) training (p < .01). Ipsilateral CME of the paretic TA (.02 ± .01 mV) and TCI (.01 ± .01 s, ipsilateral silent period; more inhibition to the lesioned M1) increased after skill (p < .05) but not strength training. Reaction time of the paretic ankle improved after skill and strength training (-.11 ± .2 and -.13 ± .20 s, respectively; p < .05) and was sustained at 60 min. No changes were observed during the sham condition. Our findings may inform future studies for using non-paretic leg movements as a priming modality, especially for those who are contraindicated to other priming paradigms (e.g., brain stimulation) or unable to perform paretic leg movements. Conclusion: Non-paretic leg movements can be used as a priming modality, especially for those who are contraindicated to other priming paradigms (e.g., brain stimulation) or unable to perform paretic leg movements.
Keywords: cortical priming; cross-education; reaction time; severe stroke; transcranial magnetic stimulation.
© 2023 The Authors. European Journal of Neuroscience published by Federation of European Neuroscience Societies and John Wiley & Sons Ltd.
Conflict of interest statement
Conflict of Interest
The authors have no conflicts of interest to declare.
Figures


References
-
- Bawa P, Chalmers GR, Stewart H, & Eisen AA (2002). Responses of ankle extensor and flexor motoneurons to transcranial magnetic stimulation. Journal of Neurophysiology, 88(1), 124–132. - PubMed
-
- Boroojerdi B, Diefenbach K, & Ferbert A (1996). Transcallosal inhibition in cortical and subcortical cerebral vascular lesions. Journal of the Neurological Sciences, 144(1–2), 160–170. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

