The Rho guanine dissociation inhibitor α inhibits skeletal muscle Rac1 activity and insulin action
- PMID: 37364105
- PMCID: PMC10318982
- DOI: 10.1073/pnas.2211041120
The Rho guanine dissociation inhibitor α inhibits skeletal muscle Rac1 activity and insulin action
Abstract
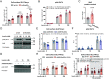
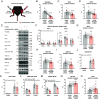
The molecular events governing skeletal muscle glucose uptake have pharmacological potential for managing insulin resistance in conditions such as obesity, diabetes, and cancer. With no current pharmacological treatments to target skeletal muscle insulin sensitivity, there is an unmet need to identify the molecular mechanisms that control insulin sensitivity in skeletal muscle. Here, the Rho guanine dissociation inhibitor α (RhoGDIα) is identified as a point of control in the regulation of insulin sensitivity. In skeletal muscle cells, RhoGDIα interacted with, and thereby inhibited, the Rho GTPase Rac1. In response to insulin, RhoGDIα was phosphorylated at S101 and Rac1 dissociated from RhoGDIα to facilitate skeletal muscle GLUT4 translocation. Accordingly, siRNA-mediated RhoGDIα depletion increased Rac1 activity and elevated GLUT4 translocation. Consistent with RhoGDIα's inhibitory effect, rAAV-mediated RhoGDIα overexpression in mouse muscle decreased insulin-stimulated glucose uptake and was detrimental to whole-body glucose tolerance. Aligning with RhoGDIα's negative role in insulin sensitivity, RhoGDIα protein content was elevated in skeletal muscle from insulin-resistant patients with type 2 diabetes. These data identify RhoGDIα as a clinically relevant controller of skeletal muscle insulin sensitivity and whole-body glucose homeostasis, mechanistically by modulating Rac1 activity.
Keywords: GLUT4 translocation; glucose uptake; insulin sensitivity; skeletal muscle; type 2 diabetes.
Conflict of interest statement
The authors declare no competing interest.
Figures





References
-
- Han X., et al. , Cancer causes metabolic perturbations associated with reduced insulin-stimulated glucose uptake in peripheral tissues and impaired muscle microvascular perfusion. Metabolism 105, 154169 (2020). - PubMed
-
- Màrmol J. M., et al. , Insulin resistance in patients with cancer: a systematic review and meta-analysis. Acta Oncol. (Madr). 62, 364–371 (2023). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

