A Label-Free Carbohydrate-Based Electrochemical Sensor to Detect Escherichia coli Pathogenic Bacteria Using D-mannose on a Glassy Carbon Electrode
- PMID: 37366984
- PMCID: PMC10296727
- DOI: 10.3390/bios13060619
A Label-Free Carbohydrate-Based Electrochemical Sensor to Detect Escherichia coli Pathogenic Bacteria Using D-mannose on a Glassy Carbon Electrode
Abstract
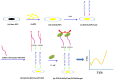
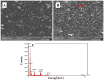
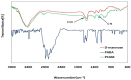
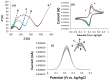
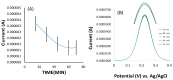
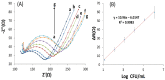
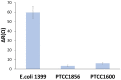
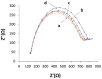
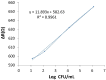
Controlling water and food contamination by pathogenic organisms requires quick, simple, and low-cost methods. Using the affinity between mannose and type I fimbriae in the cell wall of Escherichia coli (E. coli) bacteria as evaluation elements compared to the conventional plate counting technique enables a reliable sensing platform for the detection of bacteria. In this study, a simple new sensor was developed based on electrochemical impedance spectroscopy (EIS) for rapid and sensitive detection of E. coli. The biorecogniton layer of the sensor was formed by covalent attachment of p-carboxyphenylamino mannose (PCAM) to gold nanoparticles (AuNPs) electrodeposited on the surface of a glassy carbon electrode (GCE). The resultant structure of PCAM was characterized and confirmed using a Fourier Transform Infrared Spectrometer (FTIR). The developed biosensor demonstrated a linear response with a logarithm of bacterial concentration (R2 = 0.998) in the range of 1.3 × 10 1~1.3 × 106 CFU·mL-1 with the limit of detection of 2 CFU·mL-1 within 60 min. The sensor did not generate any significant signals with two non-target strains, demonstrating the high selectivity of the developed biorecognition chemistry. The selectivity of the sensor and its applicability to analysis of the real samples were investigated in tap water and low-fat milk samples. Overall, the developed sensor showed to be promising for the detection of E. coli pathogens in water and low-fat milk due to its high sensitivity, short detection time, low cost, high specificity, and user-friendliness.
Keywords: adhesion FimH; electrochemical impedance spectroscopy; electrochemical sensing; electrodeposited; gold nanoparticles.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Label-free and ultrasensitive electrochemical transferrin detection biosensor based on a glassy carbon electrode and gold nanoparticles.Int J Biol Macromol. 2024 Jan;256(Pt 1):128312. doi: 10.1016/j.ijbiomac.2023.128312. Epub 2023 Nov 22. Int J Biol Macromol. 2024. PMID: 38000589
-
Reduced graphene oxide/nile blue/gold nanoparticles complex-modified glassy carbon electrode used as a sensitive and label-free aptasensor for ratiometric electrochemical sensing of dopamine.Anal Chim Acta. 2018 Sep 26;1025:154-162. doi: 10.1016/j.aca.2018.03.036. Epub 2018 Mar 29. Anal Chim Acta. 2018. PMID: 29801604
-
A glassy carbon electrode modified with reduced graphene oxide and gold nanoparticles for electrochemical aptasensing of lipopolysaccharides from Escherichia coli bacteria.Mikrochim Acta. 2019 Nov 15;186(12):787. doi: 10.1007/s00604-019-3957-9. Mikrochim Acta. 2019. PMID: 31732807
-
Electrochemical and optical biosensors for the detection of E. Coli.Clin Chim Acta. 2025 Jan 15;565:119984. doi: 10.1016/j.cca.2024.119984. Epub 2024 Oct 12. Clin Chim Acta. 2025. PMID: 39401653 Review.
-
Carbon Dots in the Detection of Pathogenic Bacteria and Viruses.Crit Rev Anal Chem. 2024;54(2):219-246. doi: 10.1080/10408347.2022.2072168. Epub 2022 May 9. Crit Rev Anal Chem. 2024. PMID: 35533107 Review.
Cited by
-
Harnessing glycofluoroforms for impedimetric biosensing.Chem Sci. 2024 Sep 13;15(39):16086-95. doi: 10.1039/d4sc04409f. Online ahead of print. Chem Sci. 2024. PMID: 39282644 Free PMC article.
-
Comb-Tipped Coupled Cantilever Sensor for Enhanced Real-Time Detection of E. coli Bacteria.Sensors (Basel). 2025 Jul 3;25(13):4145. doi: 10.3390/s25134145. Sensors (Basel). 2025. PMID: 40648400 Free PMC article.
-
Surface Functionalization and Escherichia coli Detection Using Surface-Enhanced Raman Spectroscopy Driven by Functional Organic Polymer/Gold Nanofilm-Based Microfluidic Chip.Biosensors (Basel). 2023 Nov 21;13(12):994. doi: 10.3390/bios13120994. Biosensors (Basel). 2023. PMID: 38131754 Free PMC article.
References
-
- Doyle M.P. Food Safety: Bacterial Contamination. In: Caballero B., editor. Encyclopedia of Human Nutrition. 3rd ed. Academic Press; Waltham, MA, USA: 2013. pp. 322–330.
-
- Thomas L.V., Rowe B., McConnell M.M. In strains of Escherichia coli O167 a single plasmid encodes for the coli surface antigens CS5 and CS6 of putative colonization factor PCF8775, heat-stable enterotoxin, and colicin Ia. Infect. Immun. 1987;55:1929–1931. doi: 10.1128/iai.55.8.1929-1931.1987. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

