From Synthesis to Clinical Trial: Novel Bioinductive Calcium Deficient HA/β-TCP Bone Grafting Nanomaterial
- PMID: 37368306
- PMCID: PMC10326822
- DOI: 10.3390/nano13121876
From Synthesis to Clinical Trial: Novel Bioinductive Calcium Deficient HA/β-TCP Bone Grafting Nanomaterial
Abstract
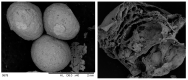
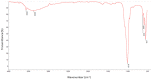
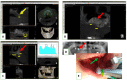
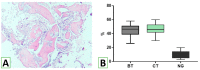
Maxillary sinus augmentation is a commonly used procedure for the placement of dental implants. However, the use of natural and synthetic materials in this procedure has resulted in postoperative complications ranging from 12% to 38%. To address this issue, we developed a novel calcium deficient HA/β-TCP bone grafting nanomaterial using a two-step synthesis method with appropriate structural and chemical parameters for sinus lifting applications. We demonstrated that our nanomaterial exhibits high biocompatibility, enhances cell proliferation, and stimulates collagen expression. Furthermore, the degradation of β-TCP in our nanomaterial promotes blood clot formation, which supports cell aggregation and new bone growth. In a clinical trial involving eight cases, we observed the formation of compact bone tissue 8 months after the operation, allowing for the successful installation of dental implants without any early postoperative complications. Our results suggest that our novel bone grafting nanomaterial has the potential to improve the success rate of maxillary sinus augmentation procedures.
Keywords: HA/β-TCP; bionanomaterials; bionanotechnology; bone grafting nanomaterials; clinical cases; dental implants; maxillary sinus lifting; nanostructures; scaffolds; synthetic bone materials.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







Similar articles
-
Sinus augmentation using human mesenchymal stem cells loaded into a beta-tricalcium phosphate/hydroxyapatite scaffold.Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008 Aug;106(2):203-9. doi: 10.1016/j.tripleo.2007.12.001. Epub 2008 Apr 18. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008. PMID: 18424115
-
Promoting Osseointegration of Dental Implants in Dog Maxillary Sinus Floor Augmentation Using Dentin Matrix Protein 1-Transduced Bone Marrow Stem Cells.Tissue Eng Regen Med. 2020 Oct;17(5):705-715. doi: 10.1007/s13770-020-00277-1. Epub 2020 Jun 25. Tissue Eng Regen Med. 2020. PMID: 32588341 Free PMC article.
-
Morphometric and histomorphometric evaluations of high-purity macro/microporous beta-tricalcium phosphate in maxillary sinus floor elevation: preliminary results on a retrospective, multi-center, observational study.BMC Oral Health. 2021 Sep 16;21(1):448. doi: 10.1186/s12903-021-01797-5. BMC Oral Health. 2021. PMID: 34530808 Free PMC article.
-
A Human Clinical, Histological, Histomorphometrical, and Radiographical Study on Biphasic HA-Beta-TCP 30/70 in Maxillary Sinus Augmentation.Clin Implant Dent Relat Res. 2015 Jun;17(3):610-8. doi: 10.1111/cid.12145. Epub 2013 Sep 4. Clin Implant Dent Relat Res. 2015. PMID: 24004190
-
Augmentation grafting of the maxillary sinus for placement of dental implants: anatomy, physiology, and procedures.Implant Dent. 1999;8(1):36-46. doi: 10.1097/00008505-199901000-00004. Implant Dent. 1999. PMID: 10356455 Review.
Cited by
-
Periodontal ligament stem cell tissue engineering scaffolds can guide and promote canine periodontal tissue regeneration.Front Vet Sci. 2024 Oct 9;11:1465879. doi: 10.3389/fvets.2024.1465879. eCollection 2024. Front Vet Sci. 2024. PMID: 39444741 Free PMC article.
-
Clinical efficacy of allograft bone combined with calcium phosphate bone powder in repairing bone defects of benign bone tumors: A retrospective controlled study.BMC Surg. 2025 Mar 14;25(1):99. doi: 10.1186/s12893-025-02823-x. BMC Surg. 2025. PMID: 40087717 Free PMC article.
References
-
- Pesce P., Menini M., Canullo L., Khijmatgar S., Modenese L., Gallifante G., Del Fabbro M. Radiographic and Histomorphometric Evaluation of Biomaterials Used for Lateral Sinus Augmentation: A Systematic Review on the Effect of Residual Bone Height and Vertical Graft Size on New Bone Formation and Graft Shrinkage. J. Clin. Med. 2021;10:4996. doi: 10.3390/jcm10214996. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

