Enfortumab Vedotin With or Without Pembrolizumab in Cisplatin-Ineligible Patients With Previously Untreated Locally Advanced or Metastatic Urothelial Cancer
- PMID: 37369081
- PMCID: PMC10852367
- DOI: 10.1200/JCO.22.02887
Enfortumab Vedotin With or Without Pembrolizumab in Cisplatin-Ineligible Patients With Previously Untreated Locally Advanced or Metastatic Urothelial Cancer
Abstract
Purpose: Patients with locally advanced or metastatic urothelial cancer (la/mUC) who are ineligible for cisplatin-based therapy have limited first-line (1L) treatment options and significant need for improved therapies. Enfortumab vedotin (EV) and pembrolizumab (Pembro) individually have shown a survival benefit in urothelial cancer in second-line + la/mUC settings. Here, we present data from the pivotal trial of EV plus Pembro (EV + Pembro) in the 1L setting.
Patients and methods: In Cohort K of the EV-103 phase Ib/II study, cisplatin-ineligible patients with previously untreated la/mUC were randomly assigned 1:1 to receive EV as monotherapy or in combination with Pembro. The primary end point was confirmed objective response rate (cORR) per blinded independent central review. Secondary end points included duration of response (DOR) and safety. There were no formal statistical comparisons between treatment arms.
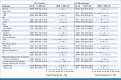
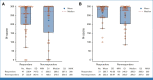
Results: The cORR was 64.5% (95% CI, 52.7 to 75.1) and 45.2% (95% CI, 33.5 to 57.3) for patients treated with EV + Pembro (N = 76) and EV monotherapy (N = 73), respectively. The median DOR was not reached for the combination and was 13.2 months for monotherapy; 65.4% and 56.3% of patients who responded to the combination and monotherapy, respectively, maintained a response at 12 months. The most common grade 3 or higher treatment-related adverse events (TRAEs) in patients treated with the combination were maculopapular rash (17.1%), fatigue (9.2%), and neutropenia (9.2%). EV TRAEs of special interest (any grade) in the combination arm included skin reactions (67.1%) and peripheral neuropathy (60.5%).
Conclusion: EV + Pembro showed a high cORR with durable responses as 1L treatment in cisplatin-ineligible patients with la/mUC. Patients who received EV monotherapy had a response and safety profile consistent with previous studies. Adverse events for EV + Pembro were manageable, with no new safety signals observed.
Trial registration: ClinicalTrials.gov NCT03288545.
Conflict of interest statement
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
Figures






Comment in
-
Combinations, Sequencing, and the Contribution of Components: New Frontline Standards for Metastatic Urothelial Carcinoma.J Clin Oncol. 2023 Sep 1;41(25):4084-4086. doi: 10.1200/JCO.23.00987. Epub 2023 Jul 25. J Clin Oncol. 2023. PMID: 37490639 No abstract available.
References
-
- Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–E386. - PubMed
-
- Shah MV, McGovern A, Hepp Z. Targeted literature review of the burden of illness in urothelial carcinoma. Value Health. 2018;21:S32–S33.
-
- Dash A, Galsky MD, Vickers AJ, et al. Impact of renal impairment on eligibility for adjuvant cisplatin-based chemotherapy in patients with urothelial carcinoma of the bladder. Cancer. 2006;107:506–513. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

