The ACSL4 Network Regulates Cell Death and Autophagy in Diseases
- PMID: 37372148
- PMCID: PMC10295397
- DOI: 10.3390/biology12060864
The ACSL4 Network Regulates Cell Death and Autophagy in Diseases
Abstract
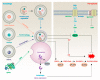
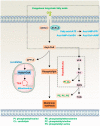
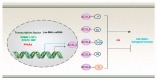
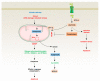
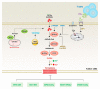
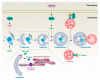
Lipid metabolism, cell death, and autophagy are interconnected processes in cells. Dysregulation of lipid metabolism can lead to cell death, such as via ferroptosis and apoptosis, while lipids also play a crucial role in the regulation of autophagosome formation. An increased autophagic response not only promotes cell survival but also causes cell death depending on the context, especially when selectively degrading antioxidant proteins or organelles that promote ferroptosis. ACSL4 is an enzyme that catalyzes the formation of long-chain acyl-CoA molecules, which are important intermediates in the biosynthesis of various types of lipids. ACSL4 is found in many tissues and is particularly abundant in the brain, liver, and adipose tissue. Dysregulation of ACSL4 is linked to a variety of diseases, including cancer, neurodegenerative disorders, cardiovascular disease, acute kidney injury, and metabolic disorders (such as obesity and non-alcoholic fatty liver disease). In this review, we introduce the structure, function, and regulation of ACSL4; discuss its role in apoptosis, ferroptosis, and autophagy; summarize its pathological function; and explore the potential implications of targeting ACSL4 in the treatment of various diseases.
Keywords: ACSL4; apoptosis; autophagy; cancer; ferroptosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources

