Recent Advances in the Green Synthesis of Active N-Heterocycles and Their Biological Activities
- PMID: 37375820
- PMCID: PMC10304377
- DOI: 10.3390/ph16060873
Recent Advances in the Green Synthesis of Active N-Heterocycles and Their Biological Activities
Abstract
N-heterocyclic scaffolds represent a privileged architecture in the process of drug design and development. It has widespread occurrence in synthetic and natural products, either those that are established or progressing as potent drug candidates. Additionally, numerous novel N-heterocyclic analogues with remarkable physiological significance and extended pharmaceutical applications are escalating progressively. Hence, the classical synthetic protocols need to be improvised according to modern requirements for efficient and eco-friendly approaches. Numerous methodologies and technologies emerged to address the green and sustainable production of various pharmaceutically and medicinally important N-heterocyclic compounds in last few years. In this context, the current review unveils greener alternatives for direct access to categorically differentiated N-heterocyclic derivatives and its application in the establishment of biologically active potent molecules for drug design. The green and sustainable methods accentuated in this review includes microwave-assisted reactions, solvent-free approaches, heterogeneous catalysis, ultrasound reactions, and biocatalysis.
Keywords: N-heterocyclic compounds; bioactivity; drug design and development; green synthesis; pharmaceutical activity.
Conflict of interest statement
There is no conflict of interest or known competing financial interest that could have appeared to influence the work reported in this paper.
Figures


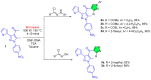




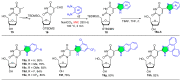
























References
-
- Baiula M., Galletti P., Martelli G., Soldati R., Belvisi L., Civera M., Dattoli S.D., Spampinato S.M., Giacomini D. New β-Lactam Derivatives Modulate Cell Adhesion and Signaling Mediated by RGD-Binding and Leukocyte Integrins. J. Med. Chem. 2016;59:9721–9742. doi: 10.1021/acs.jmedchem.6b00576. - DOI - PubMed
-
- Sun L., Huang T., Dick A., Meuser M.E., Zalloum W.A., Chen C.-H., Ding X., Gao P., Cocklin S., Lee K.-H., et al. Design, Synthesis and Structure-Activity Relationships of 4-Phenyl-1H-1,2,3-Triazole Phenylalanine Derivatives as Novel HIV-1 Capsid Inhibitors with Promising Antiviral Activities. Eur. J. Med. Chem. 2020;190:112085. doi: 10.1016/j.ejmech.2020.112085. - DOI - PMC - PubMed
-
- Zhu W.-J., Cui B.-W., Wang H.M., Nan J.-X., Piao H.-R., Lian L.-H., Jin C.H. Design, Synthesis, and Antifibrosis Evaluation of 4-(Benzo-[c][1,2,5]Thiadiazol-5-Yl)-3(5)-(6-Methyl- Pyridin-2-Yl)Pyrazole and 3(5)-(6-Methylpyridin-2-Yl)-4-(Thieno-[3,2,-c]Pyridin-2-Yl)Pyrazole Derivatives. Eur. J. Med. Chem. 2019;180:15–27. doi: 10.1016/j.ejmech.2019.07.013. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

