Different Responses to Water Deficit of Two Common Winter Wheat Varieties: Physiological and Biochemical Characteristics
- PMID: 37375865
- PMCID: PMC10304252
- DOI: 10.3390/plants12122239
Different Responses to Water Deficit of Two Common Winter Wheat Varieties: Physiological and Biochemical Characteristics
Abstract
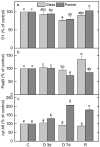
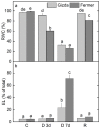
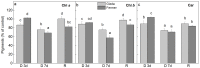
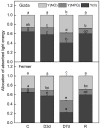
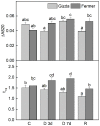
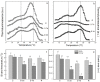
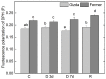
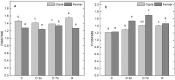
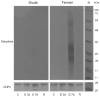
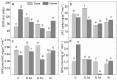
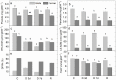
Since water scarcity is one of the main risks for the future of agriculture, studying the ability of different wheat genotypes to tolerate a water deficit is fundamental. This study examined the responses of two hybrid wheat varieties (Gizda and Fermer) with different drought resistance to moderate (3 days) and severe (7 days) drought stress, as well as their post-stress recovery to understand their underlying defense strategies and adaptive mechanisms in more detail. To this end, the dehydration-induced alterations in the electrolyte leakage, photosynthetic pigment content, membrane fluidity, energy interaction between pigment-protein complexes, primary photosynthetic reactions, photosynthetic and stress-induced proteins, and antioxidant responses were analyzed in order to unravel the different physiological and biochemical strategies of both wheat varieties. The results demonstrated that Gizda plants are more tolerant to severe dehydration compared to Fermer, as evidenced by the lower decrease in leaf water and pigment content, lower inhibition of photosystem II (PSII) photochemistry and dissipation of thermal energy, as well as lower dehydrins' content. Some of defense mechanisms by which Gizda variety can tolerate drought stress involve the maintenance of decreased chlorophyll content in leaves, increased fluidity of the thylakoid membranes causing structural alterations in the photosynthetic apparatus, as well as dehydration-induced accumulation of early light-induced proteins (ELIPs), an increased capacity for PSI cyclic electron transport and enhanced antioxidant enzyme activity (SOD and APX), thus alleviating oxidative damage. Furthermore, the leaf content of total phenols, flavonoids, and lipid-soluble antioxidant metabolites was higher in Gizda than in Fermer.
Keywords: antioxidant activity; chlorophyll fluorescence; dehydration; photosynthetic pigments and proteins; primary photosynthetic reactions; rehydration; thermoluminescence.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Hanjra M.A., Qureshi M.E. Global water crisis and future food security in an era of climate change. Food Pol. 2010;35:365–377. doi: 10.1016/j.foodpol.2010.05.006. - DOI
-
- Zhao T., Dai A. The magnitude and causes of global drought changes in the twenty-first century under a low–severe emissions scenario. J. Clim. 2015;28:4490–4512. doi: 10.1175/JCLI-D-14-00363.1. - DOI
-
- Hussain M., Farooq S., Hasan W., Ul-Allah S., Tanveer M., Farooq M., Nawaz A. Drought stress in sunflower: Physiological effects and its management through breeding and agronomic alternatives. Agric. Water Manag. 2018;201:152–166. doi: 10.1016/j.agwat.2018.01.028. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

