Clinical Activity of Combined Telomerase Vaccination and Pembrolizumab in Advanced Melanoma: Results from a Phase I Trial
- PMID: 37378632
- PMCID: PMC10425723
- DOI: 10.1158/1078-0432.CCR-23-0416
Clinical Activity of Combined Telomerase Vaccination and Pembrolizumab in Advanced Melanoma: Results from a Phase I Trial
Abstract
Purpose: Cancer vaccines represent a novel treatment modality with a complementary mode of action addressing a crucial bottleneck for checkpoint inhibitor (CPI) efficacy. CPIs are expected to release brakes in T-cell responses elicited by vaccination, leading to more robust immune responses. Increased antitumor T-cell responses may confer increased antitumor activity in patients with less immunogenic tumors, a subgroup expected to achieve reduced benefit from CPIs alone. In this trial, a telomerase-based vaccine was combined with pembrolizumab to assess the safety and clinical activity in patients with melanoma.
Patients and methods: Thirty treatment-naïve patients with advanced melanoma were enrolled. Patients received intradermal injections of UV1 with adjuvant GM-CSF at two dose levels, and pembrolizumab according to the label. Blood samples were assessed for vaccine-induced T-cell responses, and tumor tissues were collected for translational analyses. The primary endpoint was safety, with secondary objectives including progression-free survival (PFS), overall survival (OS), and objective response rate (ORR).
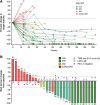
Results: The combination was considered safe and well-tolerated. Grade 3 adverse events were observed in 20% of patients, with no grade 4 or 5 adverse events reported. Vaccination-related adverse events were mostly mild injection site reactions. The median PFS was 18.9 months, and the 1- and 2-year OS rates were 86.7% and 73.3%, respectively. The ORR was 56.7%, with 33.3% achieving complete responses. Vaccine-induced immune responses were observed in evaluable patients, and inflammatory changes were detected in posttreatment biopsies.
Conclusions: Encouraging safety and preliminary efficacy were observed. Randomized phase II trials are currently ongoing.
©2023 The Authors; Published by the American Association for Cancer Research.
Figures



References
-
- Wei SC, Duffy CR, Allison JP. Fundamental mechanisms of immune checkpoint blockade therapy. Cancer Discov 2018;8:1069–86. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

