Prognostic survival biomarkers of tumor-fused dendritic cell vaccine therapy in patients with newly diagnosed glioblastoma
- PMID: 37382632
- PMCID: PMC10491709
- DOI: 10.1007/s00262-023-03482-8
Prognostic survival biomarkers of tumor-fused dendritic cell vaccine therapy in patients with newly diagnosed glioblastoma
Abstract
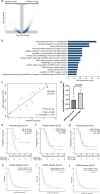
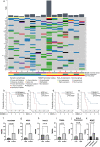
Dendritic cell (DC)-based immunotherapy has been applied to glioblastoma (GBM); however, biomarkers informing response remain poorly understood. We conducted a phase I/IIa clinical trial investigating tumor-fused DC (TFDC) immunotherapy following temozolomide-based chemoradiotherapy in patients with newly diagnosed GBM and determined prognostic factors in patients receiving TFDC immunotherapy. Twenty-eight adult patients with GBM isocitrate dehydrogenase (IDH) wild-type (IDH-WT) were enrolled; 127 TFDC vaccine injections (4.5 ± 2.6 times/patient) were administered. Patients with GBM IDH-WT had a respectable 5-year survival rate (24%), verifying the clinical activity of TFDC immunotherapy, particularly against O6-methylguanine-DNA methyltransferase (MGMT) unmethylated GBM (5-year survival rate: 33%). To identify novel factors influencing overall survival (OS) in GBM IDH-WT treated with TFDC immunotherapy, clinical parameters were assessed and comprehensive molecular profiling involving transcriptome and exome analyses was performed. MGMT promoter methylation status, extent of tumor resection, and vaccine parameters (administration frequency, DC and tumor cell numbers, and fusion ratio) were not associated with survival following TFDC immunotherapy. Old age and pre- and post-operative Karnofsky performance status were significantly correlated with OS. Low HLA-A expression and lack of CCDC88A, KRT4, TACC2, and TONSL mutations in tumor cells were correlated with better prognosis. We validated the activity of TFDC immunotherapy against GBM IDH-WT, including chemoresistant, MGMT promoter unmethylated cases. The identification of molecular biomarkers predictive of TFDC immunotherapy efficacy in GBM IDH-WT will facilitate the design of and patient stratification in a phase-3 trial to maximize treatment benefits.
Keywords: Dendritic cell; Glioblastoma; Immunotherapy; Prognostic factor; Whole tumor cell.
© 2023. The Author(s).
Conflict of interest statement
The authors have no relevant financial or non-financial interests to disclose.
Figures


References
-
- Liau LM, Ashkan K, Brem S, et al. Association of autologous tumor lysate-loaded dendritic cell vaccination with extension of survival among patients with newly diagnosed and recurrent glioblastoma: a phase 3 prospective externally controlled cohort trial. JAMA Oncol. 2023;9:112–121. doi: 10.1001/jamaoncol.2022.5370. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

