The N-degron pathway mediates lipophagy: The chemical modulation of lipophagy in obesity and NAFLD
- PMID: 37385404
- PMCID: PMC10529862
- DOI: 10.1016/j.metabol.2023.155644
The N-degron pathway mediates lipophagy: The chemical modulation of lipophagy in obesity and NAFLD
Abstract
Background and aims: Central to the pathogenesis of nonalcoholic fatty liver disease (NAFLD) is the accumulation of lipids in the liver and various fat tissues. We aimed to elucidate the mechanisms by which lipid droplets (LDs) in the liver and adipocytes are degraded by the autophagy-lysosome system and develop therapeutic means to modulate lipophagy, i.e., autophagic degradation of LDs.
Methods: We monitored the process in which LDs are pinched off by autophagic membranes and degraded by lysosomal hydrolases in cultured cells and mice. The autophagic receptor p62/SQSTM-1/Sequestosome-1 was identified as a key regulator and used as a target to develop drugs to induce lipophagy. The efficacy of p62 agonists was validated in mice to treat hepatosteatosis and obesity.
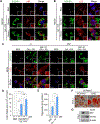
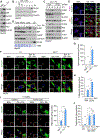
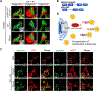
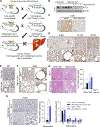
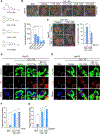
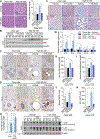
Results: We found that the N-degron pathway modulates lipophagy. This autophagic degradation initiates when the molecular chaperones including BiP/GRP78, retro-translocated from the endoplasmic reticulum, is N-terminally (Nt-) arginylated by ATE1 R-transferase. The resulting Nt-arginine (Nt-Arg) binds the ZZ domain of p62 associated with LDs. Upon binding to Nt-Arg, p62 undergoes self-polymerization and recruits LC3+ phagophores to the site of lipophagy, leading to lysosomal degradation. Liver-specific Ate1 conditional knockout mice under high fat diet developed severe NAFLD. The Nt-Arg was modified into small molecule agonists to p62 that facilitate lipophagy in mice and exerted therapeutic efficacy in obesity and hepatosteatosis of wild-type but not p62 knockout mice.
Conclusions: Our results show that the N-degron pathway modulates lipophagy and provide p62 as a drug target to treat NAFLD and other diseases related with metabolic syndrome.
Keywords: Hepatosteatosis; Lipid droplet; N-terminal arginylation; Obesity; The autophagy-lysosome system; p62/SQSTM1/Sequestosome-1.
Copyright © 2023 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interest Seoul National University and AUTOTAC Bio, Inc. have filed patent applications based on this study.
Figures



References
-
- Byrne CD and Targher G, NAFLD: a multisystem disease. J Hepatol, 2015. 62(1 Suppl): p. S47–64. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

