Cardiac lipid metabolism, mitochondrial function, and heart failure
- PMID: 37392421
- PMCID: PMC10681665
- DOI: 10.1093/cvr/cvad100
Cardiac lipid metabolism, mitochondrial function, and heart failure
Abstract
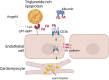
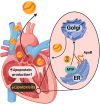
A fine balance between uptake, storage, and the use of high energy fuels, like lipids, is crucial in the homeostasis of different metabolic tissues. Nowhere is this balance more important and more precarious than in the heart. This highly energy-demanding muscle normally oxidizes almost all the available substrates to generate energy, with fatty acids being the preferred source under physiological conditions. In patients with cardiomyopathies and heart failure, changes in the main energetic substrate are observed; these hearts often prefer to utilize glucose rather than oxidizing fatty acids. An imbalance between uptake and oxidation of fatty acid can result in cellular lipid accumulation and cytotoxicity. In this review, we will focus on the sources and uptake pathways used to direct fatty acids to cardiomyocytes. We will then discuss the intracellular machinery used to either store or oxidize these lipids and explain how disruptions in homeostasis can lead to mitochondrial dysfunction and heart failure. Moreover, we will also discuss the role of cholesterol accumulation in cardiomyocytes. Our discussion will attempt to weave in vitro experiments and in vivo data from mice and humans and use several human diseases to illustrate metabolism gone haywire as a cause of or accomplice to cardiac dysfunction.
Keywords: Cholesterol; Heart failure; Lipids; Lipoprotein.
© The Author(s) 2023. Published by Oxford University Press on behalf of the European Society of Cardiology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Conflict of interest statement
Conflict of interest: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, Falk V, González-Juanatey JR, Harjola VP, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, Parissis JT, Pieske B, Riley JP, Rosano GMC, Ruilope LM, Ruschitzka F, Rutten FH, van der Meer P, Sisakian HS, Isayev E, Kurlianskaya A, Mullens W, Tokmakova M, Agathangelou P, Melenovsky V, Wiggers H, Hassanein M, Uuetoa T, Lommi J, Kostovska ES, Juilliere Y, Aladashvili A, Luchner A, Chrysohoou C, Nyolczas N, Thorgeirsson G, Weinstein JM, di Lenarda A, Aidargaliyeva N, Bajraktari G, Beishenkulov M, Kamzola G, Abdel-Massih T, Celutkiene J, Noppe S, Cassar A, Vataman E, AbirKhalil S, van Pol P, Mo R, Straburzynska-Migaj E, Fonseca C, Chioncel O, Shlyakhto E, Zavatta M, Otasevic P, Goncalvesova E, Lainscak M, Molina BD, Schaufelberger M, Suter T, Yılmaz MB, Voronkov L, Davies C. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 2016;37:2129–2200. - PubMed
-
- Kong MG, Jang SY, Jang J, Cho H-J, Lee S, Lee SE, Kim KH, Yoo B-S, Kang S-M, Baek SH, Choi D-J, Jeon E-S, Kim J-J, Cho M-C, Chae SC, Oh B-H, Lim S, Park SK, Lee H-Y. Impact of diabetes mellitus on mortality in patients with acute heart failure: a prospective cohort study. Cardiovasc Diabetol 2020;19:49. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

