This is a preprint.
Expression of RcrB confers resistance to hypochlorous acid in uropathogenic Escherichia coli
- PMID: 37398214
- PMCID: PMC10312555
- DOI: 10.1101/2023.06.01.543251
Expression of RcrB confers resistance to hypochlorous acid in uropathogenic Escherichia coli
Update in
-
Expression of RcrB confers resistance to hypochlorous acid in uropathogenic Escherichia coli.J Bacteriol. 2023 Oct 26;205(10):e0006423. doi: 10.1128/jb.00064-23. Epub 2023 Oct 4. J Bacteriol. 2023. PMID: 37791752 Free PMC article.
Abstract
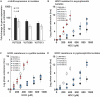
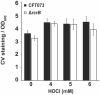
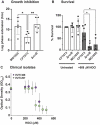
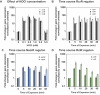
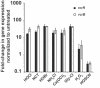
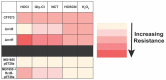
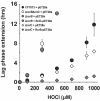
To eradicate bacterial pathogens, neutrophils are recruited to the sites of infection, where they engulf and kill microbes through the production of reactive oxygen and chlorine species (ROS/RCS). The most prominent RCS is antimicrobial oxidant hypochlorous acid (HOCl), which rapidly reacts with various amino acids side chains, including those containing sulfur and primary/tertiary amines, causing significant macromolecular damage. Pathogens like uropathogenic Escherichia coli (UPEC), the primary causative agent of urinary tract infections (UTIs), have developed sophisticated defense systems to protect themselves from HOCl. We recently identified the RcrR regulon as a novel HOCl defense strategy in UPEC. The regulon is controlled by the HOCl-sensing transcriptional repressor RcrR, which is oxidatively inactivated by HOCl resulting in the expression of its target genes, including rcrB . rcrB encodes the putative membrane protein RcrB, deletion of which substantially increases UPEC's susceptibility to HOCl. However, many questions regarding RcrB's role remain open including whether (i) the protein's mode of action requires additional help, (ii) rcrARB expression is induced by physiologically relevant oxidants other than HOCl, and (iii) expression of this defense system is limited to specific media and/or cultivation conditions. Here, we provide evidence that RcrB expression is sufficient to E. coli 's protection from HOCl and induced by and protects from several RCS but not from ROS. RcrB plays a protective role for RCS-stressed planktonic cells under various growth and cultivation conditions but appears to be irrelevant for UPEC's biofilm formation.
Importance: Bacterial infections pose an increasing threat to human health exacerbating the demand for alternative treatment options. UPEC, the most common etiological agent of urinary tract infections (UTIs), are confronted by neutrophilic attacks in the bladder, and must therefore be well equipped with powerful defense systems to fend off the toxic effects of RCS. How UPEC deal with the negative consequences of the oxidative burst in the neutrophil phagosome remains unclear. Our study sheds light on the requirements for the expression and protective effects of RcrB, which we recently identified as UPEC's most potent defense system towards HOCl-stress and phagocytosis. Thus, this novel HOCl-stress defense system could potentially serve as an attractive drug target to increase the body's own capacity to fight UTIs.
Figures



Similar articles
-
Expression of RcrB confers resistance to hypochlorous acid in uropathogenic Escherichia coli.J Bacteriol. 2023 Oct 26;205(10):e0006423. doi: 10.1128/jb.00064-23. Epub 2023 Oct 4. J Bacteriol. 2023. PMID: 37791752 Free PMC article.
-
Redox-Mediated Inactivation of the Transcriptional Repressor RcrR is Responsible for Uropathogenic Escherichia coli's Increased Resistance to Reactive Chlorine Species.mBio. 2022 Oct 26;13(5):e0192622. doi: 10.1128/mbio.01926-22. Epub 2022 Sep 8. mBio. 2022. PMID: 36073817 Free PMC article.
-
Bacterial Defense Systems against the Neutrophilic Oxidant Hypochlorous Acid.Infect Immun. 2020 Jun 22;88(7):e00964-19. doi: 10.1128/IAI.00964-19. Print 2020 Jun 22. Infect Immun. 2020. PMID: 32152198 Free PMC article. Review.
-
Roles of RcsA, an AhpD Family Protein, in Reactive Chlorine Stress Resistance and Virulence in Pseudomonas aeruginosa.Appl Environ Microbiol. 2020 Oct 1;86(20):e01480-20. doi: 10.1128/AEM.01480-20. Print 2020 Oct 1. Appl Environ Microbiol. 2020. PMID: 32801171 Free PMC article.
-
Surviving Reactive Chlorine Stress: Responses of Gram-Negative Bacteria to Hypochlorous Acid.Microorganisms. 2020 Aug 11;8(8):1220. doi: 10.3390/microorganisms8081220. Microorganisms. 2020. PMID: 32796669 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
