Interobserver variability studies in diagnostic imaging: a methodological systematic review
- PMID: 37399082
- PMCID: PMC10392644
- DOI: 10.1259/bjr.20220972
Interobserver variability studies in diagnostic imaging: a methodological systematic review
Abstract
Objectives: To review the methodology of interobserver variability studies; including current practice and quality of conducting and reporting studies.
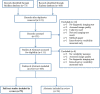
Methods: Interobserver variability studies between January 2019 and January 2020 were included; extracted data comprised of study characteristics, populations, variability measures, key results, and conclusions. Risk of bias was assessed using the COSMIN tool for assessing reliability and measurement error.
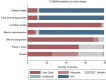
Results: Seventy-nine full-text studies were included covering various imaging tests and clinical areas. The median number of patients was 47 (IQR:23-88), and observers were 4 (IQR:2-7), with sample size justified in 12 (15%) studies. Most studies used static images (n = 75, 95%), where all observers interpreted images for all patients (n = 67, 85%). Intraclass correlation coefficients (ICC) (n = 41, 52%), Kappa (κ) statistics (n = 31, 39%) and percentage agreement (n = 15, 19%) were most commonly used. Interpretation of variability estimates often did not correspond with study conclusions. The COSMIN risk of bias tool gave a very good/adequate rating for 52 studies (66%) including any studies that used variability measures listed in the tool. For studies using static images, some study design standards were not applicable and did not contribute to the overall rating.
Conclusions: Interobserver variability studies have diverse study designs and methods, the impact of which requires further evaluation. Sample size for patients and observers was often small without justification. Most studies report ICC and κ values, which did not always coincide with the study conclusion. High ratings were assigned to many studies using the COSMIN risk of bias tool, with certain standards scored 'not applicable' when static images were used.
Advances in knowledge: The sample size for both patients and observers was often small without justification. For most studies, observers interpreted static images and did not evaluate the process of acquiring the imaging test, meaning it was not possible to assess many COSMIN risk of bias standards for studies with this design. Most studies reported intraclass correlation coefficient and κ statistics; study conclusions often did not correspond with results.
Figures

References
-
- Sardanelli F, Di Leo G. Biostatistics for Radiologists . Milano: Springer Science & Business Media; 2009. doi: 10.1007/978-88-470-1133-5 - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

