The influence of biological DMARDs on aseptic arthroplasty loosening: a retrospective cohort study
- PMID: 37402609
- PMCID: PMC10986799
- DOI: 10.1093/rheumatology/kead304
The influence of biological DMARDs on aseptic arthroplasty loosening: a retrospective cohort study
Abstract
Objective: To investigate whether biological DMARDs affect the risk of aseptic loosening after total hip/knee arthroplasty (THA/TKA) in patients with RA.
Methods: We retrospectively identified all patients suffering from RA who underwent THA/TKA at our academic centre between 2002 and 2015 and linked them with an existing prospective observational RA database at our institution. The risk of aseptic loosening was estimated using radiological signs of component loosening (RCL). A time-dependent Cox regression analysis was used to compare the risk of implant loosening between patients treated with traditional DMARDS and biological DMARDs, or alternately both over time.
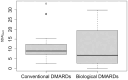
Results: A total of 155 consecutive total joint arthroplasties (TJAs) (103 TKA vs 52 THA) was retrospectively included in the study. Mean age at implantation was 59 ± 13 years. Mean follow-up time was 69 ± 43 months. Overall, 48 (31%) TJAs showed signs of RCL, with 28 (27.2%) RCLs occurring after TKA compared with 20 after THA (38.5%). A significant difference regarding the incidence of RCL between the traditional DMARDs group (39 cases of RCL, 35%) and the biological DMARDs group (nine cases of RCL, 21%) (P = 0.026) was observed using the log-rank test. This was also true when using a time-dependent Cox regression with therapy as well as arthroplasty location (hip vs knee) as variables (P = 0.0447).
Conclusion: Biological DMARDs may reduce the incidence of aseptic loosening after TJA in patients with RA compared with traditional DMARDs. This effect seems to be more pronounced after TKA than THA.
Keywords: RA; aseptic loosening; biological DMARDs; total hip arthroplasty; total knee arthroplasty.
© The Author(s) 2023. Published by Oxford University Press on behalf of the British Society for Rheumatology.
Figures




References
-
- Smolen JS, Aletaha D, Barton A. et al. Rheumatoid arthritis. Nat Rev Dis Primers 2018;4:18001. - PubMed
-
- Emery P. Evidence supporting the benefit of early intervention in rheumatoid arthritis. J Rheumatol Suppl 2002;66:3–8. - PubMed
-
- Korpela M, Laasonen L, Hannonen P. et al. Retardation of joint damage in patients with early rheumatoid arthritis by initial aggressive treatment with disease-modifying antirheumatic drugs: five-year experience from the FIN-RACo study. Arthritis Rheum 2004;50:2072–81. - PubMed
-
- Smolen JS, Han C, Bala M. et al. Evidence of radiographic benefit of treatment with infliximab plus methotrexate in rheumatoid arthritis patients who had no clinical improvement: a detailed subanalysis of data from the anti-tumor necrosis factor trial in rheumatoid arthritis with concomitant therapy study. Arthritis Rheum 2005;52:1020–30. - PubMed
-
- Lipsky PE, van der Heijde DM, St Clair EW. et al. Infliximab and methotrexate in the treatment of rheumatoid arthritis. Anti-Tumor Necrosis Factor Trial in Rheumatoid Arthritis with Concomitant Therapy Study Group. N Engl J Med 2000;343:1594–602. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

