Extracellular tau stimulates phagocytosis of living neurons by activated microglia via Toll-like 4 receptor-NLRP3 inflammasome-caspase-1 signalling axis
- PMID: 37402829
- PMCID: PMC10319744
- DOI: 10.1038/s41598-023-37887-3
Extracellular tau stimulates phagocytosis of living neurons by activated microglia via Toll-like 4 receptor-NLRP3 inflammasome-caspase-1 signalling axis
Abstract
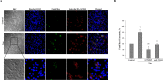
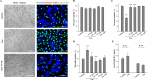
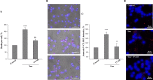
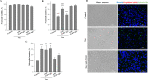
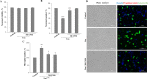
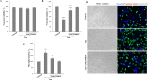
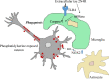
In tauopathies, abnormal deposition of intracellular tau protein followed by gradual elevation of tau in cerebrospinal fluids and neuronal loss has been documented, however, the mechanism how actually neurons die under tau pathology is largely unknown. We have previously shown that extracellular tau protein (2N4R isoform) can stimulate microglia to phagocytose live neurons, i.e. cause neuronal death by primary phagocytosis, also known as phagoptosis. Here we show that tau protein induced caspase-1 activation in microglial cells via 'Toll-like' 4 (TLR4) receptors and neutral sphingomyelinase. Tau-induced neuronal loss was blocked by caspase-1 inhibitors (Ac-YVAD-CHO and VX-765) as well as by TLR4 antibodies. Inhibition of caspase-1 by Ac-YVAD-CHO prevented tau-induced exposure of phosphatidylserine on the outer leaflet of neuronal membranes and reduced microglial phagocytic activity. We also show that suppression of NLRP3 inflammasome, which is down-stream of TLR4 receptors and mediates caspase-1 activation, by a specific inhibitor (MCC550) also prevented tau-induced neuronal loss. Moreover, NADPH oxidase is also involved in tau-induced neurotoxicity since neuronal loss was abolished by its pharmacological inhibitor. Overall, our data indicate that extracellular tau protein stimulates microglia to phagocytose live neurons via Toll-like 4 receptor-NLRP3 inflammasome-caspase-1 axis and NADPH oxidase, each of which may serve as a potential molecular target for pharmacological treatment of tauopathies.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

