Primary resistance to nivolumab plus ipilimumab therapy in patients with metastatic renal cell carcinoma
- PMID: 37403728
- PMCID: PMC10501267
- DOI: 10.1002/cam4.6306
Primary resistance to nivolumab plus ipilimumab therapy in patients with metastatic renal cell carcinoma
Abstract
Background: Nivolumab plus ipilimumab (NIVO+IPI) is the first-line treatment for patients with metastatic renal cell carcinoma (mRCC). Approximately 40% of patients achieve a durable response; however, 20% develop primary resistant disease (PRD) to NIVO+IPI, about which little is known in patients with mRCC. Therefore, this investigation aimed to evaluate the clinical implication of PRD in patients with mRCC to select better candidates in whom NIVO+IPI can be initiated as first-line therapy.
Methods: This multi-institutional retrospective cohort study used data collected between August 2015 and January 2023. In total, 120 patients with mRCC treated with NIVO+IPI were eligible. Associations between immune-related adverse events and progression-free survival, overall survival (OS), and objective response rate were analyzed. The relationship between other clinical factors and outcomes was also evaluated.
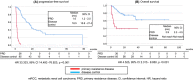
Results: The median observation period was 16 months (interquartile range, 5-27). The median age at NIVO+IPI initiation was 68 years in the male-dominant population (n = 86, 71.7%), and most patients had clear cell histology (n = 104, 86.7%). PRD was recorded in 26 (23.4%) of 111 investigated patients during NIVO+IPI therapy. Patients who experienced PRD showed worse OS (hazard ratio: 4.525, 95% confidence interval [CI]: 2.315-8.850, p < 0.001). Multivariable analysis showed that lymph node metastasis (LNM) (odds ratio: 4.274, 95% CI: 1.075-16.949, p = 0.039) was an independent risk factor for PRD.
Conclusions: PRD was strongly correlated with worse survival rates. LNM was independently associated with PRD in patients with mRCC receiving NIVO+IPI as first-line therapy and might indicate that a candidate will not benefit from NIVO+IPI.
Keywords: ipilimumab; lymph node metastasis; neoplasm metastasis; nivolumab; renal cell carcinoma; survival rate.
© 2023 The Authors. Cancer Medicine published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors have no conflicts of interest.
Figures
Similar articles
-
The lymphocyte-to-monocyte ratio as a significant inflammatory marker associated with survival of patients with metastatic renal cell carcinoma treated using nivolumab plus ipilimumab therapy.Int J Clin Oncol. 2024 Jul;29(7):1019-1026. doi: 10.1007/s10147-024-02538-8. Epub 2024 May 26. Int J Clin Oncol. 2024. PMID: 38797782
-
Primary resistance to nivolumab plus ipilimumab therapy affects second-line treatment outcomes in patients with metastatic renal cell carcinoma.Cancer Sci. 2025 Feb;116(2):444-452. doi: 10.1111/cas.16326. Epub 2024 Nov 17. Cancer Sci. 2025. PMID: 39550694 Free PMC article.
-
First-line Immuno-Oncology Combination Therapies in Metastatic Renal-cell Carcinoma: Results from the International Metastatic Renal-cell Carcinoma Database Consortium.Eur Urol. 2019 Dec;76(6):861-867. doi: 10.1016/j.eururo.2019.07.048. Epub 2019 Aug 22. Eur Urol. 2019. PMID: 31445844 Free PMC article.
-
Risks and benefits of reinduction ipilimumab/nivolumab in melanoma patients previously treated with ipilimumab/nivolumab.J Immunother Cancer. 2021 Oct;9(10):e003395. doi: 10.1136/jitc-2021-003395. J Immunother Cancer. 2021. PMID: 34702752 Free PMC article. Review.
-
Evaluating the efficacy and safety of nivolumab and ipilimumab combination therapy compared to nivolumab monotherapy in advanced cancers (excluding melanoma): a systemic review and meta-analysis.J Egypt Natl Canc Inst. 2024 May 6;36(1):14. doi: 10.1186/s43046-024-00218-2. J Egypt Natl Canc Inst. 2024. PMID: 38705953
Cited by
-
Natural Compounds in Cancer Therapy: Revealing the Role of Flavonoids in Renal Cell Carcinoma Treatment.Biomolecules. 2025 Apr 25;15(5):620. doi: 10.3390/biom15050620. Biomolecules. 2025. PMID: 40427513 Free PMC article. Review.
-
Combined Administration of Escitalopram Oxalate and Nivolumab Exhibits Synergistic Growth-Inhibitory Effects on Liver Cancer Cells through Inducing Apoptosis.Int J Mol Sci. 2023 Aug 10;24(16):12630. doi: 10.3390/ijms241612630. Int J Mol Sci. 2023. PMID: 37628813 Free PMC article.
-
The role of RNA-modifying proteins in renal cell carcinoma.Cell Death Dis. 2024 Mar 19;15(3):227. doi: 10.1038/s41419-024-06479-y. Cell Death Dis. 2024. PMID: 38503745 Free PMC article. Review.
-
Analysis of Early Progression in Advanced Renal Cell Carcinoma Treated With Nivolumab Plus Ipilimumab.Cancer Diagn Progn. 2025 May 3;5(3):344-352. doi: 10.21873/cdp.10446. eCollection 2025 May-Jun. Cancer Diagn Progn. 2025. PMID: 40322204 Free PMC article.
-
The lymphocyte-to-monocyte ratio as a significant inflammatory marker associated with survival of patients with metastatic renal cell carcinoma treated using nivolumab plus ipilimumab therapy.Int J Clin Oncol. 2024 Jul;29(7):1019-1026. doi: 10.1007/s10147-024-02538-8. Epub 2024 May 26. Int J Clin Oncol. 2024. PMID: 38797782
References
-
- Ljungberg B, Albiges L, Abu‐Ghanem Y, et al. European Association of Urology guidelines on renal cell carcinoma: the 2022 update. Eur Urol. 2022;82:399‐410. - PubMed
-
- Motzer RJ, Escudier B, McDermott DF, et al. Survival outcomes and independent response assessment with nivolumab plus ipilimumab versus sunitinib in patients with advanced renal cell carcinoma: 42‐month follow‐up of a randomized phase 3 clinical trial. J Immunother Cancer. 2020;8:e000891. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous