Temporal Changes in Nuclear Envelope Permeability during Semi-Closed Mitosis in Dictyostelium Amoebae
- PMID: 37408214
- PMCID: PMC10216430
- DOI: 10.3390/cells12101380
Temporal Changes in Nuclear Envelope Permeability during Semi-Closed Mitosis in Dictyostelium Amoebae
Abstract
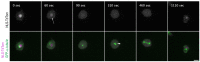
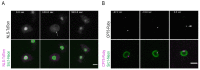
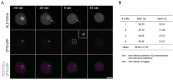
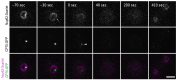
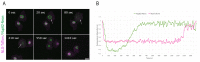
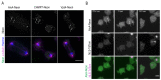
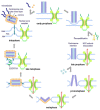
The Amoebozoan Dictyostelium discoideum exhibits a semi-closed mitosis in which the nuclear membranes remain intact but become permeabilized to allow tubulin and spindle assembly factors to access the nuclear interior. Previous work indicated that this is accomplished at least by partial disassembly of nuclear pore complexes (NPCs). Further contributions by the insertion process of the duplicating, formerly cytosolic, centrosome into the nuclear envelope and nuclear envelope fenestrations forming around the central spindle during karyokinesis were discussed. We studied the behavior of several Dictyostelium nuclear envelope, centrosomal, and nuclear pore complex (NPC) components tagged with fluorescence markers together with a nuclear permeabilization marker (NLS-TdTomato) by live-cell imaging. We could show that permeabilization of the nuclear envelope during mitosis occurs in synchrony with centrosome insertion into the nuclear envelope and partial disassembly of nuclear pore complexes. Furthermore, centrosome duplication takes place after its insertion into the nuclear envelope and after initiation of permeabilization. Restoration of nuclear envelope integrity usually occurs long after re-assembly of NPCs and cytokinesis has taken place and is accompanied by a concentration of endosomal sorting complex required for transport (ESCRT) components at both sites of nuclear envelope fenestration (centrosome and central spindle).
Keywords: Dictyostelium; centrosome; mitosis; nuclear envelope; nuclear pore complex.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Nuclear Envelope Dynamics in Dictyostelium Amoebae.Cells. 2025 Jan 26;14(3):186. doi: 10.3390/cells14030186. Cells. 2025. PMID: 39936978 Free PMC article. Review.
-
Partial Disassembly of the Nuclear Pore Complex Proteins during Semi-Closed Mitosis in Dictyostelium discoideum.Cells. 2022 Jan 25;11(3):407. doi: 10.3390/cells11030407. Cells. 2022. PMID: 35159217 Free PMC article.
-
Dictyostelium spastin is involved in nuclear envelope dynamics during semi-closed mitosis.Nucleus. 2022 Dec;13(1):144-154. doi: 10.1080/19491034.2022.2047289. Nucleus. 2022. PMID: 35298348 Free PMC article.
-
Nuclear envelope organization in Dictyostelium discoideum.Int J Dev Biol. 2019;63(8-9-10):509-519. doi: 10.1387/ijdb.190184rg. Int J Dev Biol. 2019. PMID: 31840788 Review.
-
Nucleocytoplasmic protein translocation during mitosis in the social amoebozoan Dictyostelium discoideum.Biol Rev Camb Philos Soc. 2015 Feb;90(1):126-41. doi: 10.1111/brv.12100. Epub 2014 Mar 12. Biol Rev Camb Philos Soc. 2015. PMID: 24618050 Review.
Cited by
-
A Conserved Disruption of the Nuclear Permeability Barrier in Meiosis is Controlled by a Kinase-Phosphatase Pair in Saccharomyces cerevisiae.bioRxiv [Preprint]. 2025 May 15:2025.05.14.654091. doi: 10.1101/2025.05.14.654091. bioRxiv. 2025. PMID: 40463192 Free PMC article. Preprint.
-
Nuclear Envelope Dynamics in Dictyostelium Amoebae.Cells. 2025 Jan 26;14(3):186. doi: 10.3390/cells14030186. Cells. 2025. PMID: 39936978 Free PMC article. Review.
-
Not just binary: embracing the complexity of nuclear division dynamics.Nucleus. 2024 Dec;15(1):2360601. doi: 10.1080/19491034.2024.2360601. Epub 2024 Jun 6. Nucleus. 2024. PMID: 38842147 Free PMC article. Review.
-
Dynamic Mitotic Localization of the Centrosomal Kinases CDK1, Plk, AurK, and Nek2 in Dictyostelium amoebae.Cells. 2024 Sep 10;13(18):1513. doi: 10.3390/cells13181513. Cells. 2024. PMID: 39329697 Free PMC article.
References
-
- Stafstrom J.P., Staehelin L.A. Dynamics of the Nuclear Envelope and of Nuclear Pore Complexes during Mitosis in the Drosophila Embryo. Eur. J. Cell Biol. 1984;34:179–189. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

