Elucidation of Substantial Differences in Ring-Opening Polymerization Outcomes from Subtle Variation of Glucose Carbonate-Based Monomer Substitution Patterns and Substituent Types
- PMID: 37409894
- PMCID: PMC10863030
- DOI: 10.1021/jacs.3c03339
Elucidation of Substantial Differences in Ring-Opening Polymerization Outcomes from Subtle Variation of Glucose Carbonate-Based Monomer Substitution Patterns and Substituent Types
Abstract
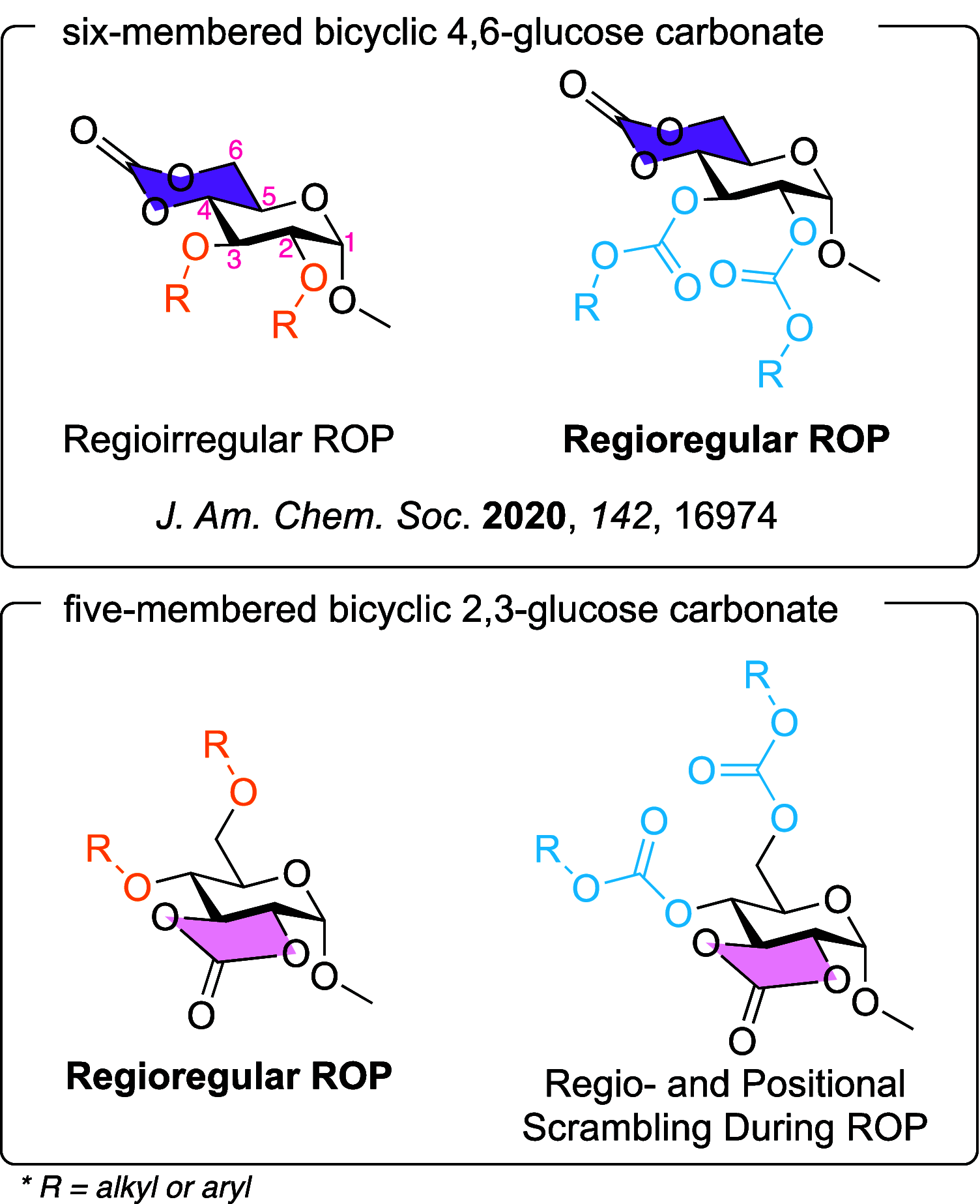
The substituents present upon five-membered bicyclic glucose carbonate monomers were found to greatly affect the reactivities and regioselectivities during ring-opening polymerization (ROP), which contrast in significant and interesting ways from previous studies on similar systems, while also leading to predictable effects on the thermal properties of the resulting polycarbonates. Polymerization behaviors were probed for a series of five five-membered bicyclic 2,3-glucose-carbonate monomers having 4,6-ether, -carbonate, or -sulfonyl urethane protecting groups, under catalysis with three different organobase catalysts. Irrespective of the organobase catalyst employed, regioregular polycarbonates were obtained via ROP of monomers with ether substituents, while the backbone connectivities of polymers derived from monomers with carbonate protecting groups suffered transcarbonylation reactions, resulting in irregular backbone connectivities and broad molar mass distributions. The sulfonyl urethane-protected monomers were unable to undergo organobase-catalyzed ROP, possibly due to the acidity of the proton in urethane functionality. The thermal behaviors of polycarbonates with ether and carbonate pendant groups were investigated in terms of thermal stability and glass transition temperature (Tg). A two-stage thermal decomposition was observed when tert-butyloxycarbonyl (BOC) groups were employed as protecting side chains, while all other polycarbonates presented high thermal stabilities with a single-stage thermal degradation. Tg was greatly affected by side-chain bulkiness, with values ranging from 39 to 139 °C. These fundamental findings of glucose-based polycarbonates may facilitate the development of next-generation sustainable highly functional materials.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Complexities of Regioselective Ring-Opening vs Transcarbonylation-Driven Structural Metamorphosis during Organocatalytic Polymerizations of Five-Membered Cyclic Carbonate Glucose Monomers.JACS Au. 2022 Jan 14;2(2):515-521. doi: 10.1021/jacsau.1c00545. eCollection 2022 Feb 28. JACS Au. 2022. PMID: 35253000 Free PMC article.
-
Invoking Side-Chain Functionality for the Mediation of Regioselectivity during Ring-Opening Polymerization of Glucose Carbonates.J Am Chem Soc. 2020 Oct 7;142(40):16974-16981. doi: 10.1021/jacs.0c05610. Epub 2020 Sep 23. J Am Chem Soc. 2020. PMID: 32965109
-
Advancing the Development of Highly-Functionalizable Glucose-Based Polycarbonates by Tuning of the Glass Transition Temperature.J Am Chem Soc. 2018 Nov 28;140(47):16053-16057. doi: 10.1021/jacs.8b10675. Epub 2018 Nov 12. J Am Chem Soc. 2018. PMID: 30418768
-
Overview: Polycarbonates via Ring-Opening Polymerization, Differences between Six- and Five-Membered Cyclic Carbonates: Inspiration for Green Alternatives.Polymers (Basel). 2022 May 16;14(10):2031. doi: 10.3390/polym14102031. Polymers (Basel). 2022. PMID: 35631913 Free PMC article. Review.
-
Lipase-catalyzed ring-opening polymerization of natural compound-based cyclic monomers.Chem Commun (Camb). 2023 Jul 25;59(60):9182-9194. doi: 10.1039/d3cc02114a. Chem Commun (Camb). 2023. PMID: 37431654 Review.
Cited by
-
Benzoylation of Tetrols: A Comparison of Regioselectivity Patterns for O- and S-Glycosides of d-Galactose.J Org Chem. 2024 Oct 4;89(19):14090-14097. doi: 10.1021/acs.joc.4c01508. Epub 2024 Sep 12. J Org Chem. 2024. PMID: 39265180 Free PMC article.
References
-
- Carafa R. N.; Foucher D. A.; Sacripante G. G. Biobased polymers from lignocellulosic sources. Green Chem. Lett. Rev. 2023, 16, 215308710.1080/17518253.2022.2153087. - DOI
-
- Haque F. M.; Ishibashi J. S. A.; Lidston C. A. L.; Shao H.; Bates F. S.; Chang A. B.; Coates G. W.; Cramer C. J.; Dauenhauer P. J.; Dichtel W. R.; et al. Defining the Macromolecules of Tomorrow through Synergistic Sustainable Polymer Research. Chem. Rev. 2022, 122, 6322–6373. 10.1021/acs.chemrev.1c00173. - DOI - PubMed
-
- Cywar R. M.; Rorrer N. A.; Hoyt C. B.; Beckham G. T.; Chen E. Y.-X. Bio-based polymers with performance-advantaged properties. Nat. Rev. Mater. 2022, 7, 83–103. 10.1038/s41578-021-00363-3. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

