Copper-Mediated Dehydrogenative C(sp3)-H Borylation of Alkanes
- PMID: 37410056
- PMCID: PMC10360158
- DOI: 10.1021/jacs.3c02185
Copper-Mediated Dehydrogenative C(sp3)-H Borylation of Alkanes
Abstract
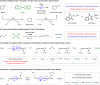
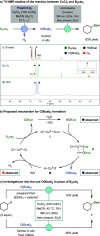
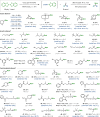
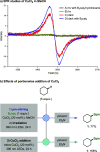
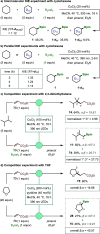
Borylations of inert carbon-hydrogen bonds are highly useful for transforming feedstock chemicals into versatile organoboron reagents. Catalysis of these reactions has historically relied on precious-metal complexes, which promote dehydrogenative borylations with diboron reagents under oxidant-free conditions. Recently, photoinduced radical-mediated borylations involving hydrogen atom transfer pathways have emerged as attractive alternatives because they provide complimentary regioselectivities and proceed under metal-free conditions. However, these net oxidative processes require stoichiometric oxidants and therefore cannot compete with the high atom economy of their precious-metal-catalyzed counterparts. Herein, we report that CuCl2 catalyzes radical-mediated, dehydrogenative C(sp3)-H borylations of alkanes with bis(catecholato)diboron under oxidant-free conditions. This is a result of an unexpected dual role of the copper catalyst, which promotes oxidation of the diboron reagent to generate an electrophilic bis-boryloxide that acts as an effective borylating agent in subsequent redox-neutral photocatalytic C-H borylations.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


References
-
- Hall D. G.Boronic Acids: Preparation and Applications in Organic Synthesis and Medicine; Wiley-VCH: Weinheim, 2005.
- Sandford C.; Aggarwal V. K. Stereospecific Functionalizations and Transformations of Secondary and Tertiary Boronic Esters. Chem. Commun. 2017, 53, 5481–5494. 10.1039/C7CC01254C. - DOI - PubMed
-
- Mkhalid I. A. I.; Barnard J. H.; Marder T. B.; Murphy J. M.; Hartwig J. F. C–H Activation for the Construction of C–B Bonds. Chem. Rev. 2010, 110, 890–931. 10.1021/cr900206p. - DOI - PubMed
- Xu L.; Wang G.-H.; Zhang S.; Wang H.; Wang L.-H.; Liu L.; Jiao J.; Li P.-F. Recent Advances in Catalytic C–H Borylation Reactions. Tetrahedron 2017, 73, 7123–7157. 10.1016/j.tet.2017.11.005. - DOI
- Bisht R.; Haldar C.; Hassan M. M. M.; Hoque M. E.; Chaturvedi J.; Chattopadhyay B. Metal-Catalysed C–H Bond Activation and Borylation. Chem. Soc. Rev. 2022, 51, 5042–5100. 10.1039/D1CS01012C. - DOI - PubMed
-
- Ohmura T.; Torigoe T.; Suginome M. Iridium-Catalysed Borylation of Sterically Hindered C(sp3)–H Bonds: Remarkable Rate Acceleration by a Catalytic Amount of Potassium tert-Butoxide. Chem. Commun. 2014, 50, 6333–6336. 10.1039/C4CC01262C. - DOI - PubMed
- Li Q.; Liskey C. W.; Hartwig J. F. Regioselective Borylation of the C–H Bonds in Alkylamines and Alkyl Ethers. Observation and Origin of High Reactivity of Primary C–H Bonds Beta to Nitrogen and Oxygen. J. Am. Chem. Soc. 2014, 136, 8755–8765. 10.1021/ja503676d. - DOI - PubMed
- Oeschger R.; Su B.; Yu I.; Ehinger C.; Romero E.; He S.; Hartwig J. F. Diverse Functionalization of Strong Alkyl C–H Bonds by Undirected Borylation. Science 2020, 368, 736–741. 10.1126/science.aba6146. - DOI - PMC - PubMed
- Jones M. R.; Fast C. D.; Schley N. D. Iridium-Catalyzed sp3 C–H Borylation in Hydrocarbon Solvent Enabled by 2,2′-Dipyridylarylmethane Ligands. J. Am. Chem. Soc. 2020, 142, 6488–6492. 10.1021/jacs.0c00524. - DOI - PubMed
- Kawazu R.; Torigoe T.; Kuninobu Y. Iridium-Catalyzed C(sp3)–H Borylation Using Silyl-Bipyridine Pincer Ligands. Angew. Chem., Int. Ed. 2022, 61, e20220232710.1002/anie.202202327. - DOI - PubMed
-
- Liskey C. W.; Hartwig J. F. Iridium-Catalyzed Borylation of Secondary C–H Bonds in Cyclic Ethers. J. Am. Chem. Soc. 2012, 134, 12422–12425. 10.1021/ja305596v. - DOI - PubMed
- Hu J.-F.; Lv J.-H.; Shi Z.-Z. Emerging Trends in C(sp3)–H Borylation. Trends Chem. 2022, 4, 685–698. 10.1016/j.trechm.2022.04.011. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

