Body fluid-derived stem cells - an untapped stem cell source in genitourinary regeneration
- PMID: 37414959
- PMCID: PMC11639537
- DOI: 10.1038/s41585-023-00787-2
Body fluid-derived stem cells - an untapped stem cell source in genitourinary regeneration
Abstract
Somatic stem cells have been obtained from solid organs and tissues, including the bone marrow, placenta, corneal stroma, periosteum, adipose tissue, dental pulp and skeletal muscle. These solid tissue-derived stem cells are often used for tissue repair, disease modelling and new drug development. In the past two decades, stem cells have also been identified in various body fluids, including urine, peripheral blood, umbilical cord blood, amniotic fluid, synovial fluid, breastmilk and menstrual blood. These body fluid-derived stem cells (BFSCs) have stemness properties comparable to those of other adult stem cells and, similarly to tissue-derived stem cells, show cell surface markers, multi-differentiation potential and immunomodulatory effects. However, BFSCs are more easily accessible through non-invasive or minimally invasive approaches than solid tissue-derived stem cells and can be isolated without enzymatic tissue digestion. Additionally, BFSCs have shown good versatility in repairing genitourinary abnormalities in preclinical models through direct differentiation or paracrine mechanisms such as pro-angiogenic, anti-apoptotic, antifibrotic, anti-oxidant and anti-inflammatory effects. However, optimization of protocols is needed to improve the efficacy and safety of BFSC therapy before therapeutic translation.
© 2023. Springer Nature Limited.
Conflict of interest statement
Competing interests
The authors declare no competing interests.
Figures

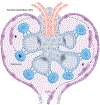

Similar articles
-
Body Fluid-Derived Stem Cells: Powering Innovative, Less-Invasive Cell Therapies.Int J Mol Sci. 2025 May 5;26(9):4382. doi: 10.3390/ijms26094382. Int J Mol Sci. 2025. PMID: 40362618 Free PMC article. Review.
-
Human placental stem cells: biomedical potential and clinical relevance.J Stem Cells. 2011;6(2):75-92. J Stem Cells. 2011. PMID: 22997848 Review.
-
Heterogeneity of proangiogenic features in mesenchymal stem cells derived from bone marrow, adipose tissue, umbilical cord, and placenta.Stem Cell Res Ther. 2016 Nov 10;7(1):163. doi: 10.1186/s13287-016-0418-9. Stem Cell Res Ther. 2016. PMID: 27832825 Free PMC article.
-
Comparison of equine bone marrow-, umbilical cord matrix and amniotic fluid-derived progenitor cells.Vet Res Commun. 2011 Feb;35(2):103-21. doi: 10.1007/s11259-010-9457-3. Epub 2010 Dec 31. Vet Res Commun. 2011. PMID: 21193959
-
Isolation of mesenchymal stem cells from human placenta: comparison with human bone marrow mesenchymal stem cells.Cell Biol Int. 2006 Sep;30(9):681-7. doi: 10.1016/j.cellbi.2006.03.009. Epub 2006 Apr 22. Cell Biol Int. 2006. PMID: 16870478
Cited by
-
Therapeutic Benefits of Stem Cells and Exosomes for Sulfur-Mustard-Induced Tissue Damage.Int J Mol Sci. 2023 Jun 9;24(12):9947. doi: 10.3390/ijms24129947. Int J Mol Sci. 2023. PMID: 37373093 Free PMC article. Review.
-
Urine-derived stem cells in neurological diseases: current state-of-the-art and future directions.Front Mol Neurosci. 2023 Oct 30;16:1229728. doi: 10.3389/fnmol.2023.1229728. eCollection 2023. Front Mol Neurosci. 2023. PMID: 37965041 Free PMC article. Review.
-
Body Fluid-Derived Stem Cells: Powering Innovative, Less-Invasive Cell Therapies.Int J Mol Sci. 2025 May 5;26(9):4382. doi: 10.3390/ijms26094382. Int J Mol Sci. 2025. PMID: 40362618 Free PMC article. Review.
-
Exploiting urine-derived induced pluripotent stem cells for advancing precision medicine in cell therapy, disease modeling, and drug testing.J Biomed Sci. 2024 May 9;31(1):47. doi: 10.1186/s12929-024-01035-4. J Biomed Sci. 2024. PMID: 38724973 Free PMC article. Review.
-
Combination of rapamycin and adipose-derived mesenchymal stromal cells enhances therapeutic potential for osteoarthritis.Stem Cell Res Ther. 2025 Jan 15;16(1):9. doi: 10.1186/s13287-024-04090-8. Stem Cell Res Ther. 2025. PMID: 39815291 Free PMC article.
References
-
- Hautmann RE et al. Functional outcome and complications following ileal neobladder reconstruction in male patients without tumor recurrence. more than 35 years of experience from a single center. J. Urol 205, 174–182 (2021). - PubMed
-
- Shekarriz B, Upadhyay J, Demirbilek S, Barthold JS & Gonzalez R Surgical complications of bladder augmentation: comparison between various enterocystoplasties in 133 patients. Urology 55, 123–128 (2000). - PubMed
-
- Lin HK et al. Biomatrices for bladder reconstruction. Adv. Drug Deliv. Rev 82–83, 47–63 (2015). - PubMed
-
- Hariharan S, Israni AK & Danovitch G Long-term survival after kidney transplantation. N. Engl. J. Med 385, 729–743 (2021). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

