A primer on modelling pancreatic islets: from models of coupled β-cells to multicellular islet models
- PMID: 37415423
- PMCID: PMC10332213
- DOI: 10.1080/19382014.2023.2231609
A primer on modelling pancreatic islets: from models of coupled β-cells to multicellular islet models
Abstract
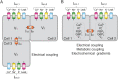
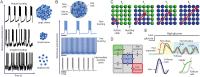
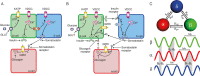
Pancreatic islets are mini-organs composed of hundreds or thousands of ɑ, β and δ-cells, which, respectively, secrete glucagon, insulin and somatostatin, key hormones for the regulation of blood glucose. In pancreatic islets, hormone secretion is tightly regulated by both internal and external mechanisms, including electrical communication and paracrine signaling between islet cells. Given its complexity, the experimental study of pancreatic islets has been complemented with computational modeling as a tool to gain a better understanding about how all the mechanisms involved at different levels of organization interact. In this review, we describe how multicellular models of pancreatic cells have evolved from the early models of electrically coupled β-cells to models in which experimentally derived architectures and both electrical and paracrine signals have been considered.
Keywords: Computational model; diabetes mellitus; gap-junctions; islets; mathematical model; paracrine.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures


Similar articles
-
δ-cells and β-cells are electrically coupled and regulate α-cell activity via somatostatin.J Physiol. 2018 Jan 15;596(2):197-215. doi: 10.1113/JP274581. Epub 2017 Nov 2. J Physiol. 2018. PMID: 28975620 Free PMC article.
-
Free fatty acid receptor 4 inhibitory signaling in delta cells regulates islet hormone secretion in mice.Mol Metab. 2021 Mar;45:101166. doi: 10.1016/j.molmet.2021.101166. Epub 2021 Jan 20. Mol Metab. 2021. PMID: 33484949 Free PMC article.
-
Gap junctions and other mechanisms of cell-cell communication regulate basal insulin secretion in the pancreatic islet.J Physiol. 2011 Nov 15;589(Pt 22):5453-66. doi: 10.1113/jphysiol.2011.218909. Epub 2011 Sep 19. J Physiol. 2011. PMID: 21930600 Free PMC article.
-
Gap junction coupling and islet delta-cell function in health and disease.Peptides. 2022 Jan;147:170704. doi: 10.1016/j.peptides.2021.170704. Epub 2021 Nov 23. Peptides. 2022. PMID: 34826505 Review.
-
Role of islet peptides in beta cell regulation and type 2 diabetes therapy.Peptides. 2018 Feb;100:212-218. doi: 10.1016/j.peptides.2017.11.014. Peptides. 2018. PMID: 29412821 Review.
Cited by
-
Deconstructing the integrated oscillator model for pancreatic β-cells.Math Biosci. 2023 Nov;365:109085. doi: 10.1016/j.mbs.2023.109085. Epub 2023 Oct 4. Math Biosci. 2023. PMID: 37802364 Free PMC article.
-
Network representation of multicellular activity in pancreatic islets: Technical considerations for functional connectivity analysis.PLoS Comput Biol. 2024 May 13;20(5):e1012130. doi: 10.1371/journal.pcbi.1012130. eCollection 2024 May. PLoS Comput Biol. 2024. PMID: 38739680 Free PMC article.
References
-
- Liao EP, Brass B, Abelev Z, Poretsky L.. Endocrine Pancreas. In: Poretsky L, editor. Principles of diabetes mellitus. Cham, Switzerland: Springer International Publishing; 2017. pp. 23–20. doi:10.1007/978-3-319-18741-9_3. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
