Separation and Washing of Candida Cells from White Blood Cells Using Viscoelastic Microfluidics
- PMID: 37420947
- PMCID: PMC10146744
- DOI: 10.3390/mi14040712
Separation and Washing of Candida Cells from White Blood Cells Using Viscoelastic Microfluidics
Abstract
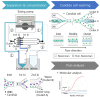
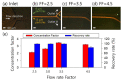
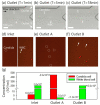
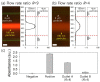
An early and accurate diagnosis of Candida albicans is critical for the rapid antifungal treatment of candidemia, a mortal bloodstream infection. This study demonstrates viscoelastic microfluidic techniques for continuous separation, concentration, and subsequent washing of Candida cells in the blood. The total sample preparation system contains two-step microfluidic devices: a closed-loop separation and concentration device and a co-flow cell-washing device. To determine the flow conditions of the closed-loop device, such as the flow rate factor, a mixture of 4 and 13 μm particles was used. Candida cells were successfully separated from the white blood cells (WBCs) and concentrated by 74.6-fold in the sample reservoir of the closed-loop system at 800 μL/min with a flow rate factor of 3.3. In addition, the collected Candida cells were washed with washing buffer (deionized water) in the microchannels with an aspect ratio of 2 at a total flow rate of 100 μL/min. Finally, Candida cells at extremely low concentrations (Ct > 35) became detectable after the removal of WBCs, the additional buffer solution in the closed-loop system (Ct = 30.3 ± 1.3), and further removal of blood lysate and washing (Ct = 23.3 ± 1.6).
Keywords: Candida; separation; viscoelastic fluid; washing; white blood cell.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Continuous On-Chip Cell Washing Using Viscoelastic Microfluidics.Micromachines (Basel). 2023 Aug 25;14(9):1658. doi: 10.3390/mi14091658. Micromachines (Basel). 2023. PMID: 37763821 Free PMC article.
-
Viscoelastic Separation and Concentration of Fungi from Blood for Highly Sensitive Molecular Diagnostics.Sci Rep. 2019 Feb 28;9(1):3067. doi: 10.1038/s41598-019-39175-5. Sci Rep. 2019. PMID: 30816161 Free PMC article.
-
High-Throughput Cell Concentration Using A Piezoelectric Pump in Closed-Loop Viscoelastic Microfluidics.Micromachines (Basel). 2021 Jun 9;12(6):677. doi: 10.3390/mi12060677. Micromachines (Basel). 2021. PMID: 34207912 Free PMC article.
-
Candida and candidaemia. Susceptibility and epidemiology.Dan Med J. 2013 Nov;60(11):B4698. Dan Med J. 2013. PMID: 24192246 Review.
-
Viscoelastic microfluidics: progress and challenges.Microsyst Nanoeng. 2020 Dec 14;6:113. doi: 10.1038/s41378-020-00218-x. eCollection 2020. Microsyst Nanoeng. 2020. PMID: 34567720 Free PMC article. Review.
Cited by
-
Continuous On-Chip Cell Washing Using Viscoelastic Microfluidics.Micromachines (Basel). 2023 Aug 25;14(9):1658. doi: 10.3390/mi14091658. Micromachines (Basel). 2023. PMID: 37763821 Free PMC article.
References
-
- Pappas P.G., Kauffman C.A., Andes D.R., Clancy C.J., Marr K.A., Ostrosky-Zeichner L., Reboli A.C., Schuster M.G., Vazquez J.A., Walsh T.J., et al. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2016;62:e1–e50. doi: 10.1093/cid/civ933. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

