Development of an α-synuclein positron emission tomography tracer for imaging synucleinopathies
- PMID: 37421950
- PMCID: PMC10527432
- DOI: 10.1016/j.cell.2023.06.004
Development of an α-synuclein positron emission tomography tracer for imaging synucleinopathies
Abstract
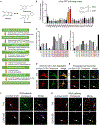
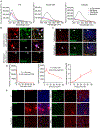
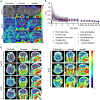
Synucleinopathies are characterized by the accumulation of α-synuclein (α-Syn) aggregates in the brain. Positron emission tomography (PET) imaging of synucleinopathies requires radiopharmaceuticals that selectively bind α-Syn deposits. We report the identification of a brain permeable and rapid washout PET tracer [18F]-F0502B, which shows high binding affinity for α-Syn, but not for Aβ or Tau fibrils, and preferential binding to α-Syn aggregates in the brain sections. Employing several cycles of counter screenings with in vitro fibrils, intraneuronal aggregates, and neurodegenerative disease brain sections from several mice models and human subjects, [18F]-F0502B images α-Syn deposits in the brains of mouse and non-human primate PD models. We further determined the atomic structure of the α-Syn fibril-F0502B complex by cryo-EM and revealed parallel diagonal stacking of F0502B on the fibril surface through an intense noncovalent bonding network via inter-ligand interactions. Therefore, [18F]-F0502B is a promising lead compound for imaging aggregated α-Syn in synucleinopathies.
Keywords: Lewy body; PET tracer; cryoelectron microscopy; protein aggregation; synucleinopathies.
Copyright © 2023 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests K.Y. is a co-founder of Shanghai Braegen Pharmaceuticals, Inc. that licensed the PET tracer patent from Emory University. No data are sponsored by the company.
Figures



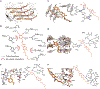
Comment in
-
Toward imaging of alpha-synuclein with PET.Cell. 2023 Aug 3;186(16):3327-3329. doi: 10.1016/j.cell.2023.06.018. Cell. 2023. PMID: 37541192
References
-
- Dev KK, Hofele K, Barbieri S, Buchman VL, and van der Putten H (2003). Part II: alpha-synuclein and its molecular pathophysiological role in neurodegenerative disease. Neuropharmacology 45, 14–44. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

