The current status of prostate cancer treatment and PSMA theranostics
- PMID: 37424944
- PMCID: PMC10328176
- DOI: 10.1177/17588359231182293
The current status of prostate cancer treatment and PSMA theranostics
Abstract
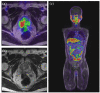
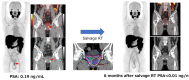
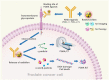
In the treatment of cancer, understanding the disease status, or accurate staging, is extremely important, and various imaging techniques are used. Computed tomography (CT), magnetic resonance imaging, and scintigrams are commonly used for solid tumors, and advances in these technologies have improved the accuracy of diagnosis. In the clinical practice of prostate cancer, CT and bone scans have been considered especially important for detecting metastases. Nowadays, CT and bone scans are called conventional methods because positron emission tomography (PET), especially prostate-specific membrane antigen (PSMA)/PET, is extremely sensitive in detecting metastases. Advances in functional imaging, such as PET, are advancing the diagnosis of cancer by allowing information to be added to the morphological diagnosis. Furthermore, PSMA is known to be upregulated depending on the malignancy of the prostate cancer grade and resistance to therapy. Therefore, it is often highly expressed in castration-resistant prostate cancer (CRPC) with poor prognosis, and its therapeutic application has been attempted for around two decades. PSMA theranostics refers to a type of cancer treatment that combines both diagnosis and therapy using a PSMA. The theranostic approach uses a radioactive substance attached to a molecule that targets PSMA protein on cancer cells. This molecule is injected into the patient's bloodstream and can be used for both imaging the cancer cells with a PET scan (PSMA PET imaging) and delivering radiation directly to the cancer cells (PSMA-targeted radioligand therapy), with the aim of minimizing damage to healthy tissue. Recently, in an international phase III trial, the impact of 177Lu-PSMA-617 therapy was studied in patients with advanced PSMA-positive metastatic CRPC who had previously been treated with specific inhibitors and regimens. The trial revealed that 177Lu-PSMA-617 significantly extended both progression-free survival and overall survival compared to standard care alone. Although there was a higher incidence of grade 3 or above adverse events with 177Lu-PSMA-617, it did not negatively impact the patients' quality of life. PSMA theranostics is currently being studied and used primarily for the treatment of prostate cancer, but it has the potential to be applied to other types of cancers as well.
Keywords: PSMA; diagnosis; prostate cancer; radiotherapy; theranostics.
© The Author(s), 2023.
Conflict of interest statement
MP and J-YS declare no relationships/activities/interests.
Figures
References
-
- Cancer Statistics. Cancer Information Service, National Cancer Center, Japan (Vital Statistics of Japan, Ministry of Health, Labour and Welfare. Cancer Statistics, 2023.
-
- Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004; 351: 1502–1512. - PubMed
-
- Fizazi K, Scher HI, Molina A, et al. Abiraterone acetate for treatment of metastatic castration-resistant prostate cancer: final overall survival analysis of the COU-AA-301 randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol 2012; 13: 983–992. - PubMed
-
- Ryan CJ, Smith MR, Fizazi K, et al. Abiraterone acetate plus prednisone versus placebo plus prednisone in chemotherapy-naive men with metastatic castration-resistant prostate cancer (COU-AA-302): final overall survival analysis of a randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol 2015; 16: 152–160. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

