Exploring novel aryl/heteroaryl-isosteres of phenylthiazole against multidrug-resistant bacteria
- PMID: 37425632
- PMCID: PMC10323310
- DOI: 10.1039/d3ra02778c
Exploring novel aryl/heteroaryl-isosteres of phenylthiazole against multidrug-resistant bacteria
Abstract
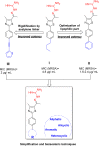
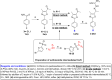
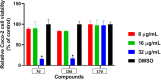
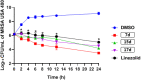
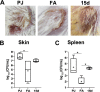
Antimicrobial resistance has become a concern as a worldwide threat. A novel scaffold of phenylthiazoles was recently evaluated against multidrug-resistant Staphylococci to control the emergence and spread of antimicrobial resistance, showing good results. Several structural modifications are needed based on the structure-activity relationships (SARs) of this new antibiotic class. Previous studies revealed the existence of two key structural features essential for the antibacterial activity, the guanidine head and lipophilic tail. In this study, a new series of twenty-three phenylthiazole derivatives were synthesized utilizing the Suzuki coupling reaction to explore the lipophilic part. The in vitro antibacterial activity was evaluated against a range of clinical isolates. The three most promising compounds, 7d, 15d and 17d, with potent MIC values against MRSA USA300 were selected for further antimicrobial evaluation. The tested compounds exhibited potent results against the tested MSSA, MRSA, and VRSA strains (concentration: 0.5 to 4 μg mL-1). Compound 15d inhibited MRSA USA400 at a concentration of 0.5 μg mL-1 (one-fold more potent than vancomycin) and showed low MIC values against ten clinical isolates, including linezolid-resistant strain MRSA NRS119 and three vancomycin-resistant isolates VRSA 9/10/12. Moreover, compound 15d retained its potent antibacterial activity using the in vivo model by the burden reduction of MRSA USA300 in skin-infected mice. The tested compounds also showed good toxicity profiles and were found to be highly tolerable to Caco-2 cells at concentrations of up to 16 μg mL-1, with 100% of the cells remaining viable.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
All authors declare that they have no conflict of interest.
Figures
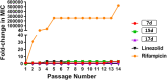
Similar articles
-
A hope for ineffective antibiotics to return to treatment: investigating the anti-biofilm potential of melittin alone and in combination with penicillin and oxacillin against multidrug resistant-MRSA and -VRSA.Front Microbiol. 2024 Feb 1;14:1269392. doi: 10.3389/fmicb.2023.1269392. eCollection 2023. Front Microbiol. 2024. PMID: 38370578 Free PMC article.
-
Selenium Nanoparticles as Candidates for Antibacterial Substitutes and Supplements against Multidrug-Resistant Bacteria.Biomolecules. 2021 Jul 14;11(7):1028. doi: 10.3390/biom11071028. Biomolecules. 2021. PMID: 34356651 Free PMC article.
-
Investigating the Antibacterial Activity of Biphenylthiazoles against Methicillin- and Vancomycin-Resistant Staphylococcus aureus (MRSA and VRSA).J Med Chem. 2017 May 11;60(9):4074-4085. doi: 10.1021/acs.jmedchem.7b00392. Epub 2017 May 1. J Med Chem. 2017. PMID: 28436655
-
Alkynyl-containing phenylthiazoles: Systemically active antibacterial agents effective against methicillin-resistant Staphylococcus aureus (MRSA).Eur J Med Chem. 2018 Mar 25;148:195-209. doi: 10.1016/j.ejmech.2018.02.031. Epub 2018 Feb 12. Eur J Med Chem. 2018. PMID: 29459278 Free PMC article.
-
The Anti-MRSA Activity of Phenylthiazoles: A Comprehensive Review.Curr Pharm Des. 2022;28(43):3469-3477. doi: 10.2174/1381612829666221124112006. Curr Pharm Des. 2022. PMID: 36424796 Review.
Cited by
-
Ligand-based discovery of novel N-arylpyrrole derivatives as broad-spectrum antimicrobial agents with antibiofilm and anti-virulence activity.J Enzyme Inhib Med Chem. 2025 Dec;40(1):2523970. doi: 10.1080/14756366.2025.2523970. Epub 2025 Jul 8. J Enzyme Inhib Med Chem. 2025. PMID: 40626382 Free PMC article.
-
Phenyltriazole-based sulfonamides: novel dual-target agents against MRSA biofilms and resistant pathogens.RSC Adv. 2025 Jun 17;15(22):17186-17202. doi: 10.1039/d5ra02412a. eCollection 2025 May 21. RSC Adv. 2025. PMID: 40529822 Free PMC article.
-
Naphthylthiazoles: a class of broad-spectrum antifungals.RSC Med Chem. 2023 Aug 29;14(10):2089-2099. doi: 10.1039/d3md00323j. eCollection 2023 Oct 18. RSC Med Chem. 2023. PMID: 37859711 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

