This is a preprint.
Tessellation of artificial touch via microstimulation of human somatosensory cortex
- PMID: 37425877
- PMCID: PMC10327055
- DOI: 10.1101/2023.06.23.545425
Tessellation of artificial touch via microstimulation of human somatosensory cortex
Abstract
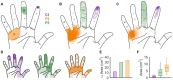
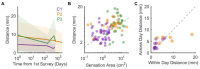
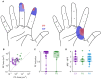
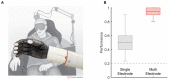
When we interact with objects, we rely on signals from the hand that convey information about the object and our interaction with it. A basic feature of these interactions, the locations of contacts between the hand and object, is often only available via the sense of touch. Information about locations of contact between a brain-controlled bionic hand and an object can be signaled via intracortical microstimulation (ICMS) of somatosensory cortex (S1), which evokes touch sensations that are localized to a specific patch of skin. To provide intuitive location information, tactile sensors on the robotic hand drive ICMS through electrodes that evoke sensations at skin locations matching sensor locations. This approach requires that ICMS-evoked sensations be focal, stable, and distributed over the hand. To systematically investigate the localization of ICMS-evoked sensations, we analyzed the projected fields (PFs) of ICMS-evoked sensations - their location and spatial extent - from reports obtained over multiple years from three participants implanted with microelectrode arrays in S1. First, we found that PFs vary widely in their size across electrodes, are highly stable within electrode, are distributed over large swaths of each participant's hand, and increase in size as the amplitude or frequency of ICMS increases. Second, while PF locations match the locations of the receptive fields (RFs) of the neurons near the stimulating electrode, PFs tend to be subsumed by the corresponding RFs. Third, multi-channel stimulation gives rise to a PF that reflects the conjunction of the PFs of the component channels. By stimulating through electrodes with largely overlapping PFs, then, we can evoke a sensation that is experienced primarily at the intersection of the component PFs. To assess the functional consequence of this phenomenon, we implemented multichannel ICMS-based feedback in a bionic hand and demonstrated that the resulting sensations are more localizable than are those evoked via single-channel ICMS.
Figures




References
-
- Bjånes D.A., Bashford L., Pejsa K., Lee B., Liu C.Y., Andersen R.A., 2022. Multi-channel intra-cortical micro-stimulation yields quick reaction times and evokes natural somatosensations in a human participant. 10.1101/2022.08.08.22278389 - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
