Characterizing Adrenergic Regulation of Glucose Transporter 4-Mediated Glucose Uptake and Metabolism in the Heart
- PMID: 37426525
- PMCID: PMC10322917
- DOI: 10.1016/j.jacbts.2022.11.008
Characterizing Adrenergic Regulation of Glucose Transporter 4-Mediated Glucose Uptake and Metabolism in the Heart
Abstract
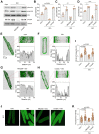
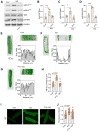
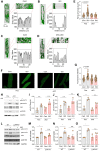
Whereas adrenergic stimulation promotes cardiac function that demands more fuel and energy, how this receptor controls cardiac glucose metabolism is not defined. This study shows that the cardiac β2 adrenoreceptor (β2AR) is required to increase glucose transporter 4 (GLUT4)-mediated glucose uptake in myocytes and glucose oxidation in working hearts via activating the cardiac β2AR and promotes the G inhibitory-phosphoinositide 3-kinase-protein kinase B cascade to increase phosphorylation of TBC1D4 (aka AS160), a Rab guanosine triphosphatase-activating protein, which is a key enzyme to mobilize GLUT4. Furthermore, deleting G-protein receptor kinase phosphorylation sites of β2AR blocked adrenergic stimulation of GLUT4-mediated glucose uptake in myocytes and hearts. This study defines a molecular pathway that controls cardiac GLUT4-mediated glucose uptake and metabolism under adrenergic stimulation.
Keywords: Akt substrate of 160 kDa; G-protein receptor kinase 2; adrenergic receptor; glucose oxidation; glucose transporter 4; glucose uptake.
© 2023 The Authors.
Conflict of interest statement
Dr Zhu has received an American Heart Association postdoctoral fellowship. Dr Li has received support from National Institutes of Health grants (R01HL158515, R01GM124108) and U.S. Department of Veterans Affairs’ VA Merit Award (I01BX005625. Dr Xiang has received support from National Institutes of Health grants (R01-HL147263, R01-HL162825) and U.S. Department of Veterans Affairs’ VA Merit Awards (01BX002900, 01BX005100); and is an established investigator of the American Heart Association. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose.
Figures


Similar articles
-
Regulation of glucose transporter 4 translocation by the Rab guanosine triphosphatase-activating protein AS160/TBC1D4: role of phosphorylation and membrane association.Mol Endocrinol. 2008 Dec;22(12):2703-15. doi: 10.1210/me.2008-0111. Epub 2008 Sep 18. Mol Endocrinol. 2008. PMID: 18801932 Free PMC article.
-
In vivo glucoregulation and tissue-specific glucose uptake in female Akt substrate 160 kDa knockout rats.PLoS One. 2020 Feb 13;15(2):e0223340. doi: 10.1371/journal.pone.0223340. eCollection 2020. PLoS One. 2020. PMID: 32053588 Free PMC article.
-
Gi-mediated translocation of GLUT4 is independent of p85/p110alpha and p110gamma phosphoinositide 3-kinases but might involve the activation of Akt kinase.Biochem J. 2000 Feb 1;345 Pt 3(Pt 3):543-55. Biochem J. 2000. PMID: 10642513 Free PMC article.
-
Role of Akt substrate of 160 kDa in insulin-stimulated and contraction-stimulated glucose transport.Appl Physiol Nutr Metab. 2007 Jun;32(3):557-66. doi: 10.1139/H07-026. Appl Physiol Nutr Metab. 2007. PMID: 17510697 Review.
-
Beta-adrenergic signaling in the heart: dual coupling of the beta2-adrenergic receptor to G(s) and G(i) proteins.Sci STKE. 2001 Oct 16;2001(104):re15. doi: 10.1126/stke.2001.104.re15. Sci STKE. 2001. PMID: 11604549 Review.
Cited by
-
At the Crossroads of Cardiac Beta-Adrenergic Receptor Signaling and Myocardial Glucose Metabolism.JACC Basic Transl Sci. 2023 Jun 26;8(6):656-657. doi: 10.1016/j.jacbts.2023.02.016. eCollection 2023 Jun. JACC Basic Transl Sci. 2023. PMID: 37426537 Free PMC article.
-
Senescence-like Phenotype After Chronic Exposure to Isoproterenol in Primary Quiescent Immune Cells.Biomolecules. 2024 Nov 28;14(12):1528. doi: 10.3390/biom14121528. Biomolecules. 2024. PMID: 39766235 Free PMC article.
-
The dual GLP-1 and GIP receptor agonist tirzapetide provides an unintended interaction with the β-adrenoceptors and plays a role in glucose metabolism in hyperglycemic or senescent cardiac cells.Cardiovasc Diabetol. 2025 Aug 18;24(1):338. doi: 10.1186/s12933-025-02828-z. Cardiovasc Diabetol. 2025. PMID: 40826463 Free PMC article.
-
β2ARs: double edge sword in heart function.Trends Mol Med. 2023 Jun;29(6):422-424. doi: 10.1016/j.molmed.2023.03.003. Epub 2023 Mar 27. Trends Mol Med. 2023. PMID: 36990857 Free PMC article.
References
-
- Neubauer S. The failing heart—an engine out of fuel. N Engl J Med. 2007;356(11):1140–1151. - PubMed
-
- Stanley W.C., Recchia F.A., Lopaschuk G.D. Myocardial substrate metabolism in the normal and failing heart. Physiol Rev. 2005;85(3):1093–1129. - PubMed
-
- Bertrand L., Auquier J., Renguet E., et al. Glucose transporters in cardiovascular system in health and disease. Pflugers Arch. 2020;472(9):1385–1399. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

