The PRO-RCC study: a long-term PROspective Renal Cell Carcinoma cohort in the Netherlands, providing an infrastructure for 'Trial within Cohorts' study designs
- PMID: 37434119
- PMCID: PMC10337109
- DOI: 10.1186/s12885-023-11094-9
The PRO-RCC study: a long-term PROspective Renal Cell Carcinoma cohort in the Netherlands, providing an infrastructure for 'Trial within Cohorts' study designs
Abstract
Background: Ongoing research in the field of both localized, locally advanced and metastatic renal cell carcinoma has resulted in the availability of multiple treatment options. Hence, many questions are still unanswered and await further research. A nationwide collaborative registry allows to collect corresponding data. For this purpose, the Dutch PROspective Renal Cell Carcinoma cohort (PRO-RCC) has been founded, for the prospective collection of long-term clinical data, patient reported outcome measures (PROMs) and patient reported experience measures (PREMs).
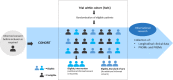
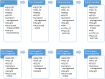
Methods: PRO-RCC is designed as a multicenter cohort for all Dutch patients with renal cell carcinoma (RCC). Recruitment will start in the Netherlands in 2023. Importantly, participants may also consent to participation in a 'Trial within cohorts' studies (TwiCs). The TwiCs design provides a method to perform (randomized) interventional studies within the registry. The clinical data collection is embedded in the Netherlands Cancer Registry (NCR). Next to the standardly available data on RCC, additional clinical data will be collected. PROMS entail Health-Related Quality of Life (HRQoL), symptom monitoring with optional ecological momentary assessment (EMA) of pain and fatigue, and optional return to work- and/or nutrition questionnaires. PREMS entail satisfaction with care. Both PROMS and PREMS are collected through the PROFILES registry and are accessible for the patient and the treating physician.
Trial registration: Ethical board approval has been obtained (2021_218) and the study has been registered at ClinicalTrials.gov (NCT05326620).
Discussion: PRO-RCC is a nationwide long-term cohort for the collection of real-world clinical data, PROMS and PREMS. By facilitating an infrastructure for the collection of prospective data on RCC, PRO-RCC will contribute to observational research in a real-world study population and prove effectiveness in daily clinical practice. The infrastructure of this cohort also enables that interventional studies can be conducted with the TwiCs design, without the disadvantages of classic RCTs such as slow patient accrual and risk of dropping out after randomization.
Keywords: Kidney cancer; Renal cell carcinoma; Urological cancer.
© 2023. The Author(s).
Conflict of interest statement
SFO reports research grants from Novartis and Celldex Therapeutics, and consultancy fees from Bristol Myers Squibb (all paid to the institution) outside the submitted work. HMW reports personal fees from Ipsen, Astellas and Roche outside the submitted work. Other authors have no competing interests to declare.
Figures
Similar articles
-
PrimerX: A Bayesian Multistage Cohort Embedded Randomised Trial to Evaluate the Role of Deferred Local Therapy of the Primary Tumour in Combination with Immune Checkpoint Inhibitor-based First-line Therapy in Metastatic Renal Cell Carcinoma Patients.Eur Urol Open Sci. 2024 Oct 14;70:28-35. doi: 10.1016/j.euros.2024.09.002. eCollection 2024 Dec. Eur Urol Open Sci. 2024. PMID: 39483517 Free PMC article.
-
Health-related Quality of Life Assessment in Renal Cell Cancer: A Scoping Review.Eur Urol Oncol. 2025 Feb;8(1):201-212. doi: 10.1016/j.euo.2024.09.007. Epub 2024 Oct 4. Eur Urol Oncol. 2025. PMID: 39366818
-
Cohort profile - the Renal cell cancer: Lifestyle, prognosis and quality of life (ReLife) study in the Netherlands.BMJ Open. 2023 Mar 27;13(3):e066909. doi: 10.1136/bmjopen-2022-066909. BMJ Open. 2023. PMID: 36972960 Free PMC article.
-
Transcriptomic signatures related to the obesity paradox in patients with clear cell renal cell carcinoma: a cohort study.Lancet Oncol. 2020 Feb;21(2):283-293. doi: 10.1016/S1470-2045(19)30797-1. Epub 2019 Dec 20. Lancet Oncol. 2020. PMID: 31870811 Free PMC article.
-
How to routinely collect data on patient-reported outcome and experience measures in renal registries in Europe: an expert consensus meeting.Nephrol Dial Transplant. 2015 Oct;30(10):1605-14. doi: 10.1093/ndt/gfv209. Epub 2015 May 16. Nephrol Dial Transplant. 2015. PMID: 25982327 Free PMC article.
Cited by
-
A National Study of the Rate of Benign Pathology After Partial Nephrectomy for T1 Renal Cell Carcinoma: Should We Be Satisfied?Cancers (Basel). 2024 Oct 17;16(20):3518. doi: 10.3390/cancers16203518. Cancers (Basel). 2024. PMID: 39456612 Free PMC article.
-
Immunotherapy in metastatic renal cell carcinoma: Insights from a Dutch nationwide cohort.Eur Urol Open Sci. 2025 Feb 10;72:42-45. doi: 10.1016/j.euros.2025.01.008. eCollection 2025 Feb. Eur Urol Open Sci. 2025. PMID: 40026657 Free PMC article.
-
PrimerX: A Bayesian Multistage Cohort Embedded Randomised Trial to Evaluate the Role of Deferred Local Therapy of the Primary Tumour in Combination with Immune Checkpoint Inhibitor-based First-line Therapy in Metastatic Renal Cell Carcinoma Patients.Eur Urol Open Sci. 2024 Oct 14;70:28-35. doi: 10.1016/j.euros.2024.09.002. eCollection 2024 Dec. Eur Urol Open Sci. 2024. PMID: 39483517 Free PMC article.
References
-
- Netherlands Cancer Registry. https://iknl.nl/en/ncr. Accessed 2 Jun 2021.
-
- Bhindi B, Abel EJ, Albiges L, Bensalah K, Boorjian SA, Daneshmand S, et al. Systematic review of the role of Cytoreductive Nephrectomy in the targeted therapy era and Beyond: an Individualized Approach to metastatic renal cell carcinoma. Eur Urol. 2019;75:111–28. doi: 10.1016/j.eururo.2018.09.016. - DOI - PubMed
-
- Yang Y. Partial Versus Radical Nephrectomy in patients with renal cell carcinoma: a systematic review and Meta-analysis. Urol J. 2020;17:109–17. - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials

