Active tuberculosis in inflammatory bowel disease patients: a case-control study
- PMID: 37435180
- PMCID: PMC10331078
- DOI: 10.1177/17562848231179871
Active tuberculosis in inflammatory bowel disease patients: a case-control study
Abstract
Background/aims: Anti-tumor necrosis factor (anti-TNF) drugs have been the mainstay therapy for moderate to severe inflammatory bowel disease (IBD) over the past 25 years. Nevertheless, these drugs are associated with serious opportunistic infections like tuberculosis (TB). Brazil is ranked among the 30 countries with the highest incidence of TB in the world. This study aimed at identifying risk factors for the development of active TB and describing clinical characteristics and outcomes in IBD patients followed at a tertiary referral center in Brazil.
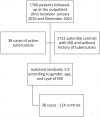
Methods: We conducted a retrospective, case-control study between January 2010 and December 2021. Active TB cases in IBD patients were randomly matched 1:3 to controls (IBD patients with no previous history of active TB) according to gender, age, and type of IBD.
Design: This was a retrospective, case-control study.
Results: A total of 38 (2.2%) cases of TB were identified from 1760 patients under regular follow-up at our outpatient clinics. Of the 152 patients included in the analysis (cases and controls), 96 (63.2%) were male, and 124 (81.6%) had Crohn's disease. Median age at TB diagnosis was 39.5 [interquartile range (IQR) 30.8-56.3]. Half of the active TB cases were disseminated (50%). Overall, 36 patients with TB (94.7%) were being treated with immunosuppressive medications. Of those, 31 (86.1%) were under anti-TNF drugs. Diagnosis of TB occurred at a median of 32 months after the first dose of anti-TNF (IQR 7-84). In multivariate analysis, IBD diagnosis older than 17 years and anti-TNF therapy were significantly associated with the development of TB (p < 0.05). After the TB treatment, 20 (52.7%) patients received anti-TNF therapy, and only one developed 'de novo' TB 10 years after the first infection.
Conclusions: TB remains a significant health problem in IBD patients from endemic regions, especially those treated with anti-TNFs. In addition, age at IBD diagnosis (>17 years old) was also a risk factor for active TB. Most cases occur after long-term therapy, suggesting a new infection. The reintroduction of anti-TNFs agents after the anti-TB treatment seems safe. These data highlight the importance of TB screening and monitoring in IBD patients living in endemic areas.
Keywords: Anti-TNF; Inflammatory bowel diseases; Tuberculosis.
© The Author(s), 2023.
Conflict of interest statement
MFCA reports receiving consulting fees from Janssen and Takeda; lecture fees from Janssen, Takeda, Pfizer, and Abbvie; and travel grant support from Takeda, Janssen, and Abbvie. LLB reports receiving lecture fees from Janssen, Takeda, and UCB and travel grant support from Takeda and Janssen. ASC reports receiving lecture fees from Janssen, Takeda, and Abbvie and travel grant support from Takeda, Janssen, and Abbvie. NSFQ reports receiving consulting payments from Janssen, lecture fees from Janssen, Takeda, UCB, and Abbvie, and travel grant support from Takeda, Janssen, and Abbvie. AOMCD reports receiving lecture fees from Janssen, Takeda, and Abbvie and travel grant support from Takeda. JO, FFJ, KSG, CAM, AZAL, and AMS have no conflict of interest to declare. No other potential conflict of interest relevant to this letter was reported.
Figures
Similar articles
-
Negative Screening Does Not Rule Out the Risk of Tuberculosis in Patients with Inflammatory Bowel Disease Undergoing Anti-TNF Treatment: A Descriptive Study on the GETAID Cohort.J Crohns Colitis. 2016 Oct;10(10):1179-85. doi: 10.1093/ecco-jcc/jjw129. Epub 2016 Jul 11. J Crohns Colitis. 2016. PMID: 27402916
-
Tuberculosis in Anti-Tumour Necrosis Factor-treated Inflammatory Bowel Disease Patients After the Implementation of Preventive Measures: Compliance With Recommendations and Safety of Retreatment.J Crohns Colitis. 2016 Oct;10(10):1186-93. doi: 10.1093/ecco-jcc/jjw022. Epub 2016 Jan 22. J Crohns Colitis. 2016. PMID: 26802085
-
Significant risk and associated factors of active tuberculosis infection in Korean patients with inflammatory bowel disease using anti-TNF agents.World J Gastroenterol. 2015 Mar 21;21(11):3308-16. doi: 10.3748/wjg.v21.i11.3308. World J Gastroenterol. 2015. PMID: 25805938 Free PMC article.
-
Visceral adiposity and inflammatory bowel disease.Int J Colorectal Dis. 2021 Nov;36(11):2305-2319. doi: 10.1007/s00384-021-03968-w. Epub 2021 Jun 9. Int J Colorectal Dis. 2021. PMID: 34104989 Review.
-
Asian Organization for Crohn's and Colitis and Asia Pacific Association of Gastroenterology consensus on tuberculosis infection in patients with inflammatory bowel disease receiving anti-tumor necrosis factor treatment. Part 2: Management.J Gastroenterol Hepatol. 2018 Jan;33(1):30-36. doi: 10.1111/jgh.14018. J Gastroenterol Hepatol. 2018. PMID: 29024102 Review.
References
-
- Maaser C, Sturm A, Vavricka SR, et al.. ECCO-ESGAR guideline for Diagnostic Assessment in IBD Part 1: Initial diagnosis, monitoring of known IBD, detection of complications. J Crohns Colitis 2019; 13: 144–164. - PubMed
-
- D’Haens GR, van Deventer S. 25 years of anti-TNF treatment for inflammatory bowel disease: lessons from the past and a look to the future. Gut 2021; 70: 1396–1405. - PubMed
-
- Lima CCG, Queiroz NSF, Sobrado CW, et al.. Critical analysis of anti-TNF use in the era of new biological agents in inflammatory bowel disease. Arq Gastroenterol 2020; 57: 323–332. - PubMed
-
- Queiroz NSF, Regueiro M. Safety considerations with biologics and new inflammatory bowel disease therapies. Curr Opin Gastroenterol 2020; 36: 257–264. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous